Researchers develop mouse model of autism spectrum disorders
Howard Hughes Medical Institute researchers have genetically engineered mice that harbor the same genetic mutation found in some people with autism and Asperger syndrome. Mice with this mutation show a similar type of social impairment and cognitive enhancement as the type seen in some people with autism spectrum disorders (ASDs). ASDs are enigmatic cognitive disorders that impair a patient's social interactions, but do not necessarily limit their intelligence.
The scientists said the mice they developed may represent an important advance in modeling autism spectrum disorders in mice and offer researchers a new tool for understanding how specific defects in neural development may lead to autism.
Howard Hughes Medical Institute investigator Thomas Südhof and his colleagues at the University of Texas Southwestern Medical Center published their findings September 6, 2007, in Science Express, which provides electronic publication of selected Science papers ahead of print.
The researchers engineered mice that have a single mutation in the gene for a protein called neuroligin-3. This protein functions as a cell adhesion molecule in synapses, the junctions that connect neurons in the brain and allow them to communicate with each other. Synapses are essential to all brain activities, such as perception, behavior, memory, and thinking. Südhof said that the neuroligin-3 mutation that his team recapitulated in the mice has been identified in some people with genetic conditions known as autism spectrum disorders (ASDs). Mutations in proteins that interact with neuroligin-3 have also been detected in some people with ASDs.
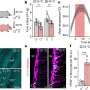
Proper function of the brain’s neuronal networks depends on a delicate balance between excitatory and inhibitory electrophysiological signaling among neurons. Südhof and his colleagues found that this balance was disrupted in the mutant mice, which also showed an increase in the signaling of inhibitory neurotransmitters. In contrast, they found that knocking out the neurologin-3 gene entirely produced no such imbalance.
The most striking behavioral abnormality they observed in the mutant mice was an impaired ability to interact socially with other mice. However, the animals showed enhanced spatial learning and memory—and were more able than normal mice to learn and to remember the location of a platform submerged in murky water.
“This combination of electrophysiological and behavioral effects is quite remarkable,” said Südhof. “It was also significant that these mice did not exhibit any other impairment of nervous system function – there was no abnormal locomotor activity or motor coordination, for example. This was a selective change, with social impairment on the one hand, yet cognitive enhancement on the other.”
Südhof said the mutant mice he and his colleagues developed potentially offer major advantages over other mouse models of ASDs. “In mouse models of autism that I am aware of, the autistic symptoms are only one minor part of the overall disease,” he said. “For example, autistic symptoms are only one component of mouse models of Fragile X syndrome.
“What sets this mouse model apart is that the mouse shows highly selective social deficits and memory enhancement, but as far as we can tell, no other pathologies. This makes it a potentially useful model for a subset of people with ASDs with just such characteristics,” he said.
Südhof his colleagues will use the mouse model to ask additional questions about the role that neuroligin proteins play in neural function and in ASDs. “We can also use these mice to study how these autistic symptoms—loss of social ability and enhanced memory—arise from the increase in inhibitory neurotransmission,” he said. “The key to understanding this mechanism will be in finding out what parts of the brain are responsible for these characteristics. And with this mouse model, we can identify precisely where the mutation acts in the brain.”
Source: Howard Hughes Medical Institute