Model of enigmatic Alzheimer's protein described for first time
Yale School of Medicine researchers have described for the first time the structure of a member in an enigmatic family of proteins linked to Alzheimer's disease, a crucial early step in understanding key mechanisms of the disease and designing drugs to combat dementia.
The technical tour de force was published online July 17 in the journal Nature.
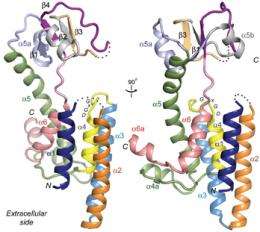
The Yale team described the crystal structure of a protease — a protein that acts like molecular scissors, cutting up proteins into component parts — found in membranes of an a single-celled organism. This membrane protein belongs to the GxGD membrane protease family. The most "famous" member of the family is a human protein called presenilin, which has been implicated in the production of amyloid beta, peptides that cause Alzheimer's disease. Presenilin carries more than 100 mutations that can lead to familial and early-onset Alzheimer's disease.
"Some of the key structural elements that allow the (archaea) protein to do its job are very similar to the existing structure models of human presenilin," noted Jian Hu, associate research scientist. Hu and colleague Yi Xue are lead authors of the Yale study.
The crystal structure of the protease called Flak from the archeon Methanococcus maripaludis is characterized by six transmembrane helices, the researchers found. Three of these segments form the catalytic site where the cutting of proteins is initiated. The arrangements of these three transmembrane helices are similar to those predicted in models of presenilin and therefore Flak is an ideal model system for presenilin research.