Gastrointestinal inflammation prevented by protein sorting factor found in cells lining the gut
The gastrointestinal tract is lined with intestinal epithelial cells (IECs) that maintain gut health by keeping bacteria and pro-inflammatory immune cells from infiltrating gut tissues. Now, a team of researchers in Japan has shown that a protein in these cells, which is responsible for sorting many proteins to particular portions of the IEC surface, plays a key role in IEC modulation of gut inflammation.
IECs are polarized cells, with a bottom surface that attaches to deeper gut tissues, and a top surface that faces the inside of the gut, where it is exposed to ingested food and gut-resident bacteria. Proteins that are created in the cell are sorted preferentially to either the top or the bottom portion of the IEC. For example, cytokine receptors are shuttled mainly to the bottom of IECs so they can respond to cytokines released by immune cells within deeper gut tissues. Led by Koji Hase at the RIKEN Research Center for Allergy and Immunology in Yokohama, the researchers thought that disruption of proper protein sorting could affect the ability of IECs to properly respond to their environment.
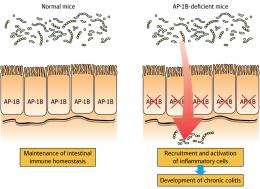
To test their theory, the researchers generated mice lacking the μ1B subunit for a sorting protein called adaptor protein-1B (AP-1B). These mice developed an inflammatory gut disease called colitis, in which large number of immune cells infiltrated the gut. Mice lacking AP-1B expressed fewer antibacterial proteins, allowing bacteria to attack gut tissue (Fig. 1). Hase and colleagues showed that this bacterial entry enhanced immune cell recruitment into the gut, because antibiotics could reduce the inflammation in these mice.
Cytokines such as interleukin-17 (IL-17) are responsible for inducing antibacterial protein expression in IECs. However, the researchers found that cells lacking AP-IB were unable to properly sort cytokine receptors, including the IL-17 receptor, to the appropriate portion of the IEC membrane. This suggested that IECs may have failed to properly respond to IL-17 because its receptors were in the wrong part of the cell.
When Hase and colleagues examined IECs in humans with an inflammatory bowel condition called Crohn’s disease, they found that expression of the μ1B subunit was reduced, and that one cytokine receptor seemed to sort to the wrong portion of the IEC surface. “AP-1B-dependent protein sorting therefore seems to control epithelial immune functions that keep the human gut healthy,” explains Hase. Enhancing the expression of μ1B could be a potential therapy for Crohn's disease, the team concludes.
More information: Takahashi, D., Hase, K., Kimura, S., Nakatsu, F., Ohmae, M., Mandai, Y., Sato, T., Date, Y., Ebisawa, M., Kato, T., et al. The epithelia-specific membrane trafficking factor AP-1B controls gut immune homeostasis in mice. Gastroenterology 141, 621–632 (2011). www.gastrojournal.org/article/S0016-5085%2811%2900611-1/abstract