Physical sciences illuminate neurodegenerative diseases

What do physicists, chemists, mathematicians and biologists have in common? One of the answers at Cambridge is a shared interest in unravelling the processes behind neurodegenerative diseases such as Alzheimer's, Parkinson's and Motor Neurone Disease.
As more people live to a ripe old age, an increasing number of us will develop neurodegenerative diseases such as Alzheimer’s. Despite the escalating economic costs and human misery associated with these diseases, we still know relatively little about how they develop or how best to tackle them.
Alzheimer’s is the most common neurodegenerative disease. “It’s an enormous problem and we’re not doing very well at the moment in slowing the disease or treating its symptoms effectively,” says Professor Peter St George-Hyslop.
Neurodegenerative diseases such as Alzheimer’s are difficult to study for several reasons. “One is that it’s not easy to get pieces of living brain,” he explains. “It’s also a disease where patients become unable to speak for themselves, so unlike people with AIDS or breast cancer they aren’t demonstrating outside the houses of Parliament demanding funding.”
Although charities and campaigners are doing sterling work raising the profile of Alzheimer’s, until recently attitudes to neurodegenerative disease had much in common with the way we viewed cancer 50 years ago.
“We are, for Alzheimer’s, like where we were for cancer in the 1950s, when people didn’t like to talk about it, were frightened or ashamed of it. And therapeutically we are in the same place; although we are beginning to learn about these diseases we don’t yet have much in the way of effective therapies,” Professor St George-Hyslop says.
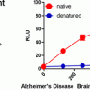
One crucial discovery is that proteins misfolding in the brain form clumps or aggregates and these play a major role in causing neurodegenerative diseases. When these proteins misfold they take on certain characteristics that become noxious to cells, but what we need to know now is why these proteins misfold, which aggregates do the damage, and how that damage occurs. Which is where physics, chemistry and mathematics enter the biological picture.
Professor St George-Hyslop leads a group of experts from disparate disciplines, each bringing different tools and different ways of working to the study of neurodegenerative diseases.
What began in late 2008 as a series of meetings has now developed into a 12-strong group funded by a £5.3 million Strategic Award from the Wellcome Trust and Medical Research Council. “It’s a very interesting group of people who came together because they wanted to come together. They each knew they had something to contribute but also that they needed something else – some skills, some knowledge, some point of view – from another member of the group,” he says.
“The biologists among us knew there were techniques that the physicists and chemists had that could help us. They in turn knew we had some biological knowledge that would help them apply, in a sensible way, their very good and insightful physical and chemical tools.”
Among the group is Professor David Klenerman from the Department of Chemistry. One of the inventors of rapid, high-throughput DNA sequencing, he is now applying this knowledge to protein misfolding. From the same department comes Professor Michele Vendruscolo, a theoretical physicist working on the mechanics and thermodynamics of protein misfolding. Professor Chris Dobson, who is also from the Department of Chemistry works on protein misfolding in neurodegenerative diseases, while from the Department of Chemical Engineering and Biotechnology Dr. Clemens Kaminski brings modern laser spectroscopy tools that allow you to watch these proteins misfold inside living cells in real time.
The group has applied these physical tools to study nematode worms in which a mutation produces the same protein misfolding that causes disease in humans. “That ability to see these things as they happen in a living model give us a much greater understanding compared with previous techniques, which essentially involved grinding up biological samples and examining them after these processes had occurred,” Professor St George-Hyslop explains.
“What’s important is the marriage of the physical tool with the biological question,” he says. And he hopes that by revealing where these misfolded proteins act, these new tools could help researchers develop ways of blocking the damage they cause in both Alzheimer’s and other neurodegenerative diseases.
“The primary goal is to understand what the beginning and the middle parts of the process are. We know what the end is – the cell dies and you get a disease – but if you know why the cells get sick and what the mechanisms are then you have a better chance of preventing or halting it,” says Professor St George- Hyslop. “Our goal is to provide that fundamental knowledge of cause and mechanism. Hopefully from that will come some idea of which parts of those pathways you can monitor as a diagnostic and which parts you can block or change as a treatment.”
More recently, the group has been enlarged by a £4.5 million grant from the National Institute of Health Research to support an extension of the Cambridge Biomedical Research Centre via the creation of a Biomedical Research Unit in Dementia for translational research. This has allowed the inclusion of researchers in immunology and in brain imaging from the Department of Medicine and the Wolfson Brain Imaging Centre.