Researchers find potential key to new treatment for mantle cell lymphoma
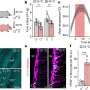
Researchers at Moffitt Cancer Center and colleagues have demonstrated that the inhibition of signal transducer and activator of transcription 3 (STAT3) in mouse models of mantle cell lymphoma (MCL), an aggressive and incurable subtype of B-cell non-Hodgkin lymphoma that becomes resistant to treatment, can harness the immune system to eradicate residual malignant cells responsible for disease relapse.
Their study appears in a recent issue of Cancer Research, published by the American Association for Cancer Research.
"Despite good initial response to first-line treatment with chemotherapy and monoclonal antibodies, almost all patients with MCL will eventually relapse," said Eduardo M. Sotomayor, M.D., a senior member at Moffitt and the Susan and John Sykes Endowed Chair for Hematologic Malignancies. "MCL has one of the worst prognoses among all B-cell non-Hodgkin lymphomas."
Moffitt researchers and colleagues have exploited evidence that suggests the manipulation of the patient's own immune system might prove a good therapeutic strategy for patients with therapy-resistant MCL.
In a study using mouse models of MCL, the researchers hypothesized that targeting and inhibiting STAT3 – a "negative regulator" of inflammatory responses in a variety of immune cells – might "unleash an effective anti-lymphoma immune response."
According to Sotomayor, much of their work is in seeking new immunotherapeutic strategies capable of breaking tolerance to tumor antigens – that is finding ways to defeat lymphoma cells' ability to resist treatment.
In this study, the researchers found that the inhibition of STAT3 in malignant B-cells, either by genetic manipulation or by using drugs (CPA-7 provided by Said M. Sebti, Ph.D., chair of the Drug Discovery Program at Moffitt and co-author of this study), render these cells "more visible" to specific immune cells (T-cells), which in turn can eradicate mantle cell lymphoma in murine models.
"The unique property of STAT3 inhibition to influence the inflammatory status of both malignant B-cells, as well as the antigen-presenting cells (as previously demonstrated by our group and others), points to pharmacologic inhibition of this signaling pathway as an appealing strategy to overcome tolerance to tumor antigens to elicit a strong anti-tumor immunity," concluded Sotomayor and his colleagues.
There have been other, similar attempts to induce an immune response, the authors noted. But these attempts offered effects that were "transient and not strong enough to fully eradicate systemic lymphoma." The authors speculated that the past limited success came from targeting either the malignant B-cell or the antigen-presenting cell, but not targeting both.
"It would be desirable to find approaches with the dual ability of enhancing the antigen-presenting function of malignant B-cells and inducing inflammatory antigen-presenting cells," Sotomayor said. "The inhibition of STAT3 signaling can do both. Therefore, STAT3 inhibition is an effective strategy in mouse models of MCL and provides a framework for future use of STAT3 inhibitors in combination with drugs that are capable of repairing defective immune responses in lymphoma patients."