Study tests potential new drug to reduce pain and symptoms of endometriosis
(Medical Xpress)—According to the Centers for Disease Control (CDC), endometriosis, a gynecological disorder in which cells from the uterus lining grow in other areas of the body, is the number one reason for a hysterectomy in women ages 18-35. Current treatment options for endometriosis are severely limited by side effects of existing medications and surgical options have more risks. A nationwide clinical trial is testing an investigational drug as a potential new option for patients with moderate to severe pain from this disease.
The Violet Petal Study, funded by Abbott and led at UC San Diego by Sanjay Agarwal, MD, FACOG, with the Department of Reproductive Medicine at UC San Diego School of Medicine, is currently recruiting patients to be part of the one to two year study to investigate the safety and effectiveness of the oral drug Elagolix. "Endometriosis can be an extremely painful disease, especially during a woman's menstrual cycle. Many women find themselves debilitated during these bouts, and the disease can also cause pain during intercourse and increase infertility," said Agarwal, director of UC San Diego Health System's Center for Endometriosis Research and Treatment (CERT), a one of a kind, multidisciplinary program in Southern California for women suffering from endometriosis.
Common therapies include injections, which bring estrogen down to very low levels, resulting in side effects such as hot flashes, bone loss and mood swings.
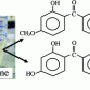
"Many patients have to take a secondary medication to counteract the side effects from current treatment options," said Agarwal. "The hope with Elagolix, taken daily, is to suppress estrogen just enough to take away the pain associated with endometriosis but without the harsh side effects to the body."
About five to 10 percent of women of reproductive age have endometriosis. The growths caused by the disease can cause tissue implants on the ovaries, bowel, rectum, bladder, and on the lining of the pelvic area. There is no cure for endometriosis and women usually need to be on treatment until menopause.
As director of CERT, Agarwal brings together a broad team of clinical and scientific experts to work with endometriosis and look at different treatment options that will improve the quality of life for patients.
"The Violet Petal Study will hopefully lead to a gentler treatment option for women living with endometriosis and advance our knowledge of this chronic disease," said Agarwal.
Eligible patients for the study must be between the ages of 18-49 and have been surgically diagnosed with endometriosis in the last seven years.
For more information on the violet petal study, please visit: www.violetpetalstudy.com/HomePage.aspx