Scientists develop novel technology to identify biomarkers for ulcerative colitis
Scientists from the Florida campus of The Scripps Research Institute have developed a novel technology that can identify, in animal models, potential biomarkers of ulcerative colitis, a type of inflammatory bowel disease that affects the lining of the colon.
The study was published October 3, 2012, in the Journal of the American Chemical Society.
The new research focuses on the protein arginine deiminases (PAD), which have been implicated in a number of diseases, including cancer, multiple sclerosis and rheumatoid arthritis. PADs participate in reactions in the body that form the amino acid citrulline in proteins through a process known as citrullination. This modification can have significant effects on the structure and function of the modified proteins.
While abnormally high PAD activity is present in a host of human diseases, the exact role of citrullination in these diseases remains unknown, largely due to the lack of readily available chemical probes to study it.
"We have developed technology to identify biomarkers for a variety of diseases in which you see abnormal PAD activity," said Paul Thompson, an associate professor in the Department of Chemistry at Scripps Research, who led the study. "This identification of potential biomarkers in animal models of ulcerative colitis is really the first step in a much larger effort. We want to push forward into rheumatoid arthritis and cancer to look for different diagnostic markers in these disease situations."
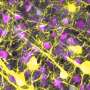
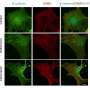
In the new study, the scientists describe a chemical probe called rhodamine‐phenylglyoxal (Rh‐PG), which tags citrulline-containing proteins with a fluorescent imaging compound.
According to Thompson, the next step will be to produce further generations of this chemical probe to isolate the biomarker proteins and determine their sites of modification, as well as to quantify the extent of the citrullination.
More information: "Seeing Citrulline: Development of a Phenylglyoxal‐Based Probe to Visualize Protein Citrullination," pubs.acs.org/doi/abs/10.1021/ja308871v