Rapid recall to fight familiar foes
The immune system's first encounter with a potential threat is a valuable learning experience. Through a process of genetic recombination, our immune B cells can potentially produce a wide array of B cell receptor (BCR) molecules, each recognizing a distinct molecular target. When a naive B cell bumps into its specific target, it initiates an immune response that yields antibodies with the same specificity as its BCR, but also gives rise to 'memory cells' that can quickly recognize the target if it appears in the future. Working with genetically modified mice, Tomohiro Kurosaki's team at the RIKEN Center for Integrative Medical Sciences in Yokohama has now gained insight into how memory cells mobilize.
"Memory B cells achieve rapid and robust antibody production during a secondary immune response, but its molecular mechanism was unknown," explains Kohei Kometani, a researcher in Kurosaki's lab and lead author of the study.
Naive B cells typically produce BCRs and antibodies that belong to the immunoglobulin M (IgM) class of proteins. In contrast, memory B cells produce an alternative immunoglobulin G (IgG) subtype of BCRs and antibodies through an additional gene recombination process called 'class switching'. Some researchers believe that the IgG form of BCR contains structural elements that stimulate the memory B cell response, while others have favored cellular mechanisms besides IgG-induced signaling.
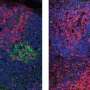
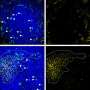
In initial experiments, the researchers worked with IgM naive and IgG memory cells that specifically recognize a known antigen, nitrophenol. As expected, the former cells proliferated in response to nitrophenol, while the latter promptly developed into antibody-secreting plasma cells, as occurs in a typical secondary immune response. To determine whether IgG is specifically responsible, Kurosaki's group generated cloned mice derived from an IgG memory cell that recognizes nitrophenol.
The resulting animals produced nitrophenol-specific IgG naive B cells, which do not normally occur in nature. Remarkably, these cells responded to nitrophenol by proliferating in essentially the same fashion as IgM naive cells, suggesting that IgG alone does not drive the memory response. A comparative analysis of gene expression revealed an alternative mechanism, controlled by a protein called Bach2. "We found that Bach2 is reduced in memory B cells, and is important for their enhanced antibody production," says Kometani.
By revealing this IgG-independent mechanism, these results should help resolve the long-standing debate over memory cell function. However, this finding is just a starting point, and Kurosaki's group is now engaged in exploring the upstream factors that switch off Bach2 production in memory cells.
More information: Kometani, K., et al. Repression of the transcription factor Bach2 contributes to predisposition of IgG1 memory B cells toward plasma cell differentiation. Immunity 39, 136–147 (2013). dx.doi.org/10.1016/j.immuni.2013.06.011