A strategy for combating drug-resistant cancers
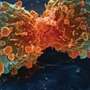
Many cancer therapies function by activating proteins like Caspase-3 (CASP3) that promote cell death. Several forms of cancer develop resistance to these drugs by down regulating CASP3 through an unknown mechanism. In the absence of CASP3, tumor cells produce another cell death promoting protein CASP7, but it is rendered inactive by the X-linked inhibitor of apoptosis protein (XIAP).
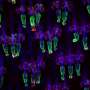
In this issue of the Journal of Clinical Investigation, Po-Huang Liang and colleagues at Academia Sinica identify a compound (I-Lys) that disrupts the interaction between CASP7 and XIAP. Release of CASP7 from this complex in drug resistant cancer cells activated cell death and reduced malignancies. Furthermore, no adverse side effects were observed in non-tumor cells treated with the drug.
In the accompanying commentary, Gregory Gores from the Mayo Clinic explains that these results are promising for combatting drug resistant cancers, but more work needs to be done to establish if I-Lys will be safe and beneficial for human use.
More information:
Targeting the XIAP/caspase-7 complex selectively kills caspase-3–deficient malignancies, J Clin Invest. 2013;123(9):3861–3875. DOI: 10.1172/JCI67951
Unshackling caspase-7 for cancer therapy, J Clin Invest. 2013;123(9):3706–3708. DOI: 10.1172/JCI71440