Simian foamy viruses readily occur between humans and macaques in urban Bangladesh

Throughout Asia, humans and monkeys live side-by side in many urban areas. An international research team from the University of Washington, Fred Hutchinson Cancer Research Center and Jahangirnagar University has been examining transmission of a virus from monkeys to humans in Bangladesh, one of the world's most densely populated countries. The scientists have found that some people in these urban areas are concurrently infected with multiple strains of simian foamy virus (SFV), including strains from more than one source (recombinant) that researchers originally detected in the monkeys.
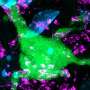
Simian foamy viruses, which are ubiquitous in nonhuman primates, are retroviruses like HIV. Retroviruses are shown to exhibit high levels of mutation and recombination – a potentially explosive combination.
Their paper, "Zoonotic simian foamy virus in Bangladesh reflects diverse patterns of transmission and co-infection" published in the Sept. 4 issue of Emerging Microbes and Infections (EMI), characterizes the simian retroviral strains that are being zoonotically transmitted and provides a glimpse into the behaviors of humans and monkeys contributing to the infections.
By analyzing what is happening at the human-primate interface, the researchers hope to protect humans from another deadly outbreak like HIV. Their focus is in Asia because it is a continent that has witnessed the emergence of several infectious diseases in the past decade. Asia also has a volatile combination of an increasingly mobile and immunocompromised population living in proximity with animals.
Since more humans have been shown to have been infected with SFV through primate contact than with any other simian-borne virus, the researchers reason that pinpointing the factors that influence SFV transmission and infection are important to a general understanding of how viruses can jump the species barrier.
"If we want to understand how, where and why these primate viruses are being transmitted, we need to be looking at SFV in Asia where millions of people and tens of thousands of macaques are interacting everyday and where we estimate that thousands of people could be infected with strains of SFV," said Lisa Jones-Engel, a primatologist with the National Primate Research Center at the University of Washington and the project leader. "These Asian rhesus macaques are Darwinian superstars. They are very responsive to change and, unlike many other species of primates, they are going to continue to thrive in human-altered habitats."
Jones-Engel said if researchers had been on the ground 50 years ago, they may have seen how simian immunodeficiency viruses (SIV) crossed the species barrier resulting in HIV.

"We have been playing catch up with the SIV-HIV question for years," she said. "We still don't know why only some viral strains are capable of establishing persistent infections in humans."
Jones-Engel said long-term surveillance is needed in the areas where humans and primates come into contact since it's unlikely that SIV/HIV will be the last primate virus to emerge into the human population.
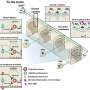
In this study, researchers collected biological samples from hundreds of people and macaques from five urban sites as well as from a group of nomadic people who travel throughout Bangladesh with their performing monkeys.
According to the paper, the towns and villages that constituted research sites for the study are likely similar to hundreds or even thousands of sites throughout Asia, where humans live alongside macaques accustomed to their presence.
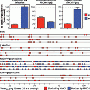
The research team found that zoonotic transmission of SFV occurred most commonly through bites. SFV replicates in oral tissues and is secreted in the saliva of infected primates. In their study, more than half of the subjects reported having being bitten at least once by a rhesus macaque, but the percentage of subjects reporting having being bitten at each site varied significantly by subjects' sex and religion. Researchers also found that primates, both human and nonhuman, can be infected with more than one strain of SFV; this is significant because co-infection may lead to viral recombination.
Among those infected with more than one strain of SFV – humans or macaques – recombination between the strains could occur.
Maxine Linial, a retrovirologist at the Fred Hutchinson Cancer Research Center (FHCRC), said successful viruses are readily transmitted and viruses evolve to be successful. She said sometimes viruses have effects on hosts to aid in transmission and these effects can have pathogenic consequences.
"Despite the fact that SFV is currently not known to be pathogenic, this was also the case for SIV before recombination and mutation allowed infection of and transmission between new hosts," Linial said. "The possibility that a pathogenic SFV strain could arise makes it essential to monitor natural infections. If a viral strain with pathogenic potential arises, we will know about it early rather than too late, which was the situation with the emergence of HIV."
By using mutations in the viruses that differentiated them from one another, the researchers were able to group the viruses into strains. They found that these strains showed a strong geographic signal, where monkeys from each given area primarily had strains characteristic of that site. However, deforestation and human transport of monkeys concentrated then moved the strains around.
"These data show a population in transition," said Frederick Matsen, a computational biologist at FHCRC. "If we were to sample 25 years earlier or 25 years later we would have seen a completely different story."
Regardless of whether SFV becomes a significant pathogen, researchers called for continued monitoring of the virus at the human and nonhuman primate interface.