Scientists think mysterious virus could be a signal of a weak immune system
More than 260,000 Americans are alive today thanks to transplant operations that have replaced their failing kidneys, hearts, lungs or livers with healthy organs donated by volunteers or accident victims.
But treatment doesn't end with surgery. Transplant recipients follow strict drug regimens designed to suppress their immune systems just enough to prevent rejection of the donated organ, but not so much as to leave them prone to infection.
Until now, maintaining this delicate balance has been something of a medical guessing game. But in a study to be published Nov. 21 in Cell, Stanford University scientists report the discovery of what may be a barometer of immune system strength: a little-known virus that proliferates as the medications suppress the immune system.
The work was led by senior author Stephen Quake, PhD, the Lee Otterson Professor in the School of Engineering and professor of bioengineering and of applied physics.
Quake and a team of collaborators, including transplant specialists from the Stanford School of Medicine, isolated specific DNA fragments from the blood of 96 heart and lung transplant patients for the study.
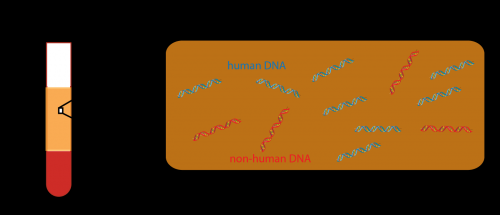
These fragments came from the blood plasma; red and white cells were extracted, leaving behind proteins and free-floating DNA. The free-floating DNA was from dead cells—the biological version of flotsam and jetsam.
Quake is a pioneer in genomics and was one of the first scientists to use genome sequencing to identify and quantify this free-floating DNA for diagnostic purposes.
One notable application of this technique relates to pregnancy. By studying fragments of cell-free DNA from maternal plasma, Quake has developed tests that can ascertain whether a woman is carrying a fetus with an extra copy of chromosome 21, which causes Down's syndrome. Additionally, cell-free DNA analysis of fragments from cancerous cells has been used to assess disease progression.
"Cell-free DNA provides an amazing window into human health, and the applications are multiplying well beyond its traditional areas of cancer and prenatal diagnostics," Quake said.
In the new study, Quake's team used these techniques to study the immune systems of transplant patients. They knew that these patients would begin taking powerful immunosuppressant drugs immediately after surgery. They reasoned that these drugs would affect the microbiome, which is the sum total of all the bacteria, viruses and fungi that inhabit the body.
Although we may not notice these microorganisms unless they cause illness, they help us digest food, excrete waste, make our feet itch or just float around with no discernable effect. In fact, the nonhuman cells that make up the microbiome "outnumber human cells in our bodies at least 10 to one," said lead author Iwijn De Vlaminck, PhD, a postdoctoral scholar in Quake's lab.
The goal of the experiment was, essentially, to use cell-free DNA analysis to perform a census of the microbiome. The hypothesis was that a systematic analysis of the nature and number of these microorganisms would reveal something about the interplay between the immune system and these nonhuman guests.
"Initially we didn't know what to expect, and we didn't know how the immunosuppressive medications would affect the numbers," De Vlaminck said.
The scientists collected plasma samples from each patient. From each sample, they extracted fragments of nonhuman DNA. They used genetic sequencing techniques to compare the fragments to a library of genetic sequences previously compiled by scientists all around the world. In this way, the team created a snapshot of the various types of bacteria, viruses and fungi in each patient's microbiome.
The scientists wanted to understand how the microbiome in each patient responded to immunosuppressant medications over time. So they took these plasma samples from each patient about seven times over a two-year period. Each time, they ran a genomic analysis of nonhuman DNA fragments in the plasma. This allowed them to chart the composition of each patient's microbiome, in a statistical version of time-lapse photography.
The scientists also studied the entire data set of 656 snapshots of all 96 patients as they looked for differences or similarities among patients.
Through this process, one stunning and unmistakable finding emerged: a mysterious micro-organism known as the anellovirus exploded into prominence as the immunosuppressive drugs kicked in, going from very low levels immediately after surgery to dominating the microbiome over time.
"It looks like the anellovirus takes advantage of the lack of immune system surveillance," De Vlaminck said.
Why or how is unknown. In fact, scientists know very little about the anellovirus. Since it was first identified in 1997, it has been found in human subjects whenever scientists have looked for its genetic fingerprints. But this common bug has not yet been identified as the cause of any disease.
But the Stanford scientists did find previous studies involving patients infected with HIV in which levels of anellovirus rose as those unfortunate patients progressed toward AIDS and the full-blown collapse of their immune systems.
These two data points—the increased prevalence of anellovirus measured in the plasma of transplant patients in the Stanford study, and the prior findings from AIDS research—provided strong hints that rising levels of anellovirus indicated a weak immune system.
But that still left a question: Was there a correlation between anellovirus levels and organ rejection?
Working with transplant specialists at the School of Medicine, the scientists looked at their data from a different angle and found out that the answer was yes. Twenty patients suffered moderate or severe episodes of organ rejection during the two years of the experiment. In these 20 patients, levels of anellovirus were "significantly lower ... at almost every point in time," the authors write, adding that "the lower viral load observed for rejecting patients is thus indicative of a higher level of immunocompetence."
Put another way, lower levels of anellovirus suggest a stronger immune system and an elevated risk of organ rejection, while higher levels of anellovirus suggest a weaker immune system with a corresponding shift in risk toward vulnerability to infection.
"We are often walking this fine line between infection and rejection, and we don't have good tools to measure immune system function for individual patients," said co-author Kiran Khush, MD, assistant professor of medicine and a cardiovascular transplant specialist at Stanford.
Instead, transplant physicians prescribe medicines according to formulas derived from factors such as time after transplant, age and weight, even though it is likely that different patients, fitting these profiles, will respond to the medications in different ways.
"We end up practicing reactive medicine," Khush said, adding, "A reliable indicator of immune system health is one of the holy grails in transplantation."
Co-senior author Hannah Valantine, MD, professor of cardiovascular medicine and senior associate dean for diversity and leadership at the School of Medicine, described the apparent linkage between anellovirus population and immune system strength as "one of the most exciting observations in my 30-year journey" of transplant research.
"These findings suggest an effective tool to individualize the monitoring and, ultimately, the treatment of rejection," she said. "In the future, this could allow us to safely lower the doses of immunosuppressive drugs patients receive, thereby avoiding devastating side effects."