Cancer drug Xeloda linked to severe skin reactions, official reports
A drug used to treat advanced breast and colorectal cancers has been linked to sometimes fatal skin reactions in patients, its Swiss manufacturer and Canadian health officials said Wednesday.
Xeloda, which treats advanced cancer after complete surgical removal, can cause "severe skin reactions," Health Canada said in a statement.
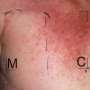
It said patients had reported severe skin reactions, such as the life-threatening skin condition known as Stevens-Johnson Syndrome or toxic epidermal necrolysis.
The signs and symptoms of this hypersensitivity include flu-like symptoms, fever, itchy skin, mouth sores, as well as painful, red or purplish skin rash that spreads and blisters, causing the top of the skin to shed, along with eye burning, itching and discharge.
Health Canada urged patients using Xeloda to contact their physician "immediately" after experiencing any of those signs or symptoms, so that they may report these adverse reactions to Roche or Health Canada.
Following requests from Canadian health authorities, Hoffmann-La Roche said it will be working with Health Canada to update the drug's prescribing information document.
It has sent a letter to healthcare professionals warning of the "risk of severe cutaneous reactions associated with the use of Xeloda," though it stressed that those cases were "very rare."
© 2013 AFP