January 10, 2014 report
Study finds progenitor cells engage in 'apical abscission' to differentiate into neurons
(Medical Xpress)—A pair of researchers working at the University of Dundee in the U.K. has discovered a new step during vertebrate neurogenesis—the process by which progenitor cells develop into neurons in embryos. In their paper published in the journal Science, Raman Das and Kate Storey describe how they used high-resolution live-cell imaging to observe early stage vertebrae brain development and in so doing discovered the previously unknown step during neurogenesis which they have called "apical abscission"—where precursor cells must leave the neural tube in which they develop to grow into different parts of the nervous system. In the same journal edition Samuel Tozer and Xavier Morin describe the process further in a Perspective piece.
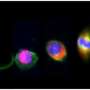
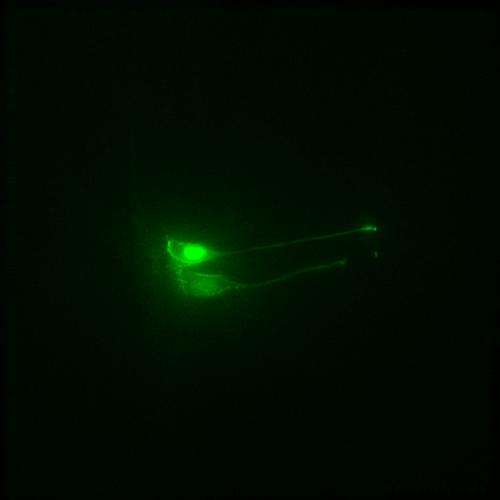
For vertebrates, neuron precursor cells develop in what is known as a neural tube. Once they reach a certain maturity level, however, they must leave the confines of the neural tube in order to make their way to the various parts of what will eventually become the organism's nervous system. Prior to this study, it was generally assumed that the migration process was no more complicated than a simple withdrawal. Instead, closer inspection by the researchers has revealed that one part of the progenitor cell undergoes an abscission (where it is severed from the rest of the cell.)
Using a very high resolution microscope, the researchers watched neurogenesis unfold in embryonic chicken embryo cells. In its beginning stages, the progenitor cell has two main parts: the primary cilium and an apical part which is connected via a tentacle-like strand of cells. As the researchers watched, one tentacle constricted, causing the two parts of the progenitor to be severed—a process the researchers have dubbed "apical abscission." The lopped off part migrated to another location and would have slowly differentiated into a neuron had it been growing in an egg. The base section, the cilium stayed behind—because it holds the signaling part of the progenitor cell, the lopped off portion was no longer able to communicate with the rest of the system, which the researchers believe is a critical part of neurogenesis.
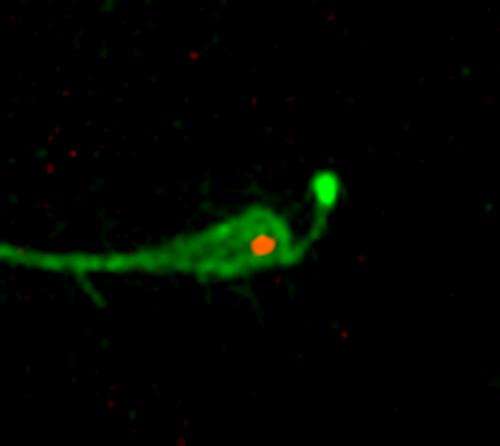
The discovery of "apical abscission" marks a major milestone in neurogenesis research and perhaps cell division in general. It's not clear if the same process occurs in other types of cells, but Tozer and Morin suggest it seems reasonable to assume it does. More research will have to be conducted to find out.
More information: Apical Abscission Alters Cell Polarity and Dismantles the Primary Cilium During Neurogenesis, Science 10 January 2014: Vol. 343 no. 6167 pp. 200-204. DOI: 10.1126/science.1247521
Abstract
Withdrawal of differentiating cells from proliferative tissue is critical for embryonic development and adult tissue homeostasis; however, the mechanisms that control this cell behavior are poorly understood. Using high-resolution live-cell imaging in chick neural tube, we uncover a form of cell subdivision that abscises apical cell membrane and mediates neuron detachment from the ventricle. This mechanism operates in chick and mouse, is dependent on actin-myosin contraction, and results in loss of apical cell polarity. Apical abscission also dismantles the primary cilium, known to transduce sonic-hedgehog signals, and is required for expression of cell-cycle-exit gene p27/Kip1. We further show that N-cadherin levels, regulated by neuronal-differentiation factor Neurog2, determine cilium disassembly and final abscission. This cell-biological mechanism may mediate such cell transitions in other epithelia in normal and cancerous conditions.
© 2014 Medical Xpress