Studies of cow antibodies help scientists understand how our own bodies work
Understanding how antibodies work is important for designing new vaccines to fight infectious diseases and certain types of cancer and for treating disorders of the immune system in animals and humans.
In research to be presented at the 58th Annual Biophysical Society Meeting, taking place in San Francisco from Feb. 15-19, Dr. Damian Ekiert, who is now at the University of California, San Francisco, will describe research he conducted as part of a team of researchers from The Scripps Research Institute in La Jolla, Calif.
In San Francisco, Ekiert will explain how the immune systems of cows are used to understand the diversity of antibodies and how that knowledge could improve the health of both people and livestock.
"First, studying the immune systems of cows and other animals helps us to understand how our own immune systems function. Second, the unique structure of these cow antibodies may be particularly well-suited for recognizing certain kinds of antigens and may be useful for antibody based therapies or diagnostics," explained Ekiert.
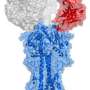
It turns out that cows make a very unusual kind of antibody different from anything scientists have ever seen before, and their antibodies are diversified by a surprising mechanism.
"Antibody diversity is particularly important because our ability to recognize and neutralize a wide range of pathogens directly depends on the diversity of our antibody repertoire—the more different kinds of antibodies we have in our bodies, the more different kinds of targets we can block," said Ekiert.
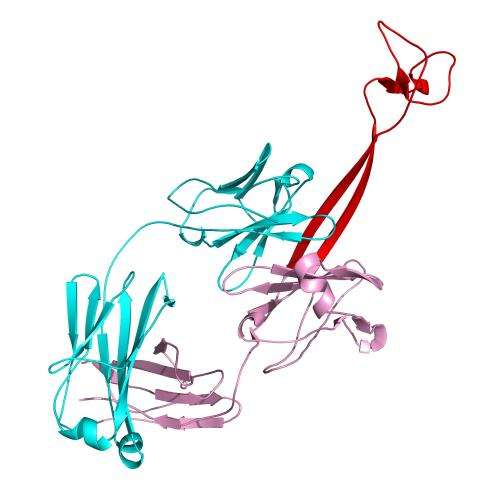
Previous work described an unusual subset of antibodies in cows that had exceptionally long loops, but no one knew what they looked like or how they were being generated. A collaborative effort including Ekiert from Ian Wilson's lab at Scripps Research as well as Feng Wang from Peter Schultz's lab and Vaughn Smider, used x-ray crystallography to determine the "ball and chain" structure of the bovine antibodies, while deep sequencing helped researchers study the function and generation of these antibodies. The next steps in realizing the potential of this research are to determine just how these antibodies recognize their target "antigen" molecules and bind to them.
In addition to the obvious benefit of helping us understand the human immune system, the research may benefit the large-scale raising of cattle, an important segment of the U.S. economy, as new vaccines can be developed to protect farm animals from common cattle diseases. Ekiert concludes, "Once we understand these mechanisms, it is possible that bovine antibodies might be able to recognize some antigens that more conventional antibodies cannot, and would help to bind, inhibit and activate targets that have thus far been intractable for antibody-based therapies."
More information: The presentation "Reshaping Antibody Diversity" by Damian C. Ekiert, Feng Wang, Ian A. Wilson, Peter G. Schultz and Vaughn V. Smider will be at 11:45 a.m. on Tuesday, February 18, 2014 in Room 303 in San Francisco's Moscone Convention Center. Abstract: tinyurl.com/k4m4twt