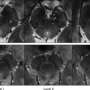
Researchers investigating genetics of Parkinson's disease
(Medical Xpress)—Researchers at Emory University School of Medicine will study individuals with genetic mutations associated with Parkinson's disease (PD) as part of the Parkinson's Progression Markers Initiative (PPMI), a large-scale biomarker study sponsored by The Michael J. Fox Foundation for Parkinson's Research.
As one of one of 32 clinical sites participating in the study, Emory will enroll participants with a known mutation of the LRRK or SNCA [alpha-synuclein] gene. Previous research has shown these mutations are associated with Parkinson's disease, and account for a greater number of PD cases among certain ethnic populations and families, notably the LRRK2 mutation in those of Ashkenazi (Eastern European) Jewish, Basque and North African Berber descent. The insight researchers gain from this study will fortify current efforts to develop a disease-modifying therapy, something that currently eludes the field.
"Studying individuals with genetic mutations associated with Parkinson's can accelerate our research toward a PD biomarker and more effective treatments," said Stewart Factor, DO, Director of the Emory Comprehensive Parkinson's Disease Center and director of the Emory Movement Disorders Center at Emory. "Although known genetic mutations currently account for only five to 10 percent of all Parkinson's cases, this population can provide invaluable information about the intricacies of the disease for all patients."
PPMI is studying clinical and imaging data and biological samples of people with a genetic mutation to identify biomarkers and speed clinical trials. PPMI will enroll 250 people with the LRRK2 mutation and Parkinson's and 250 people with the mutation but without Parkinson's. Since the SNCA mutation is rarer, the study is recruiting 50 people with Parkinson's and the mutation and 50 people with the SNCA mutation but without PD. These participants will be followed for five years. PPMI is particularly interested in testing individuals of Ashkenazi (Eastern European) Jewish descent with PD or with a relative with the disease. Interested individuals can visit www.michaeljfox.org/ppmi/genetics.
Biomarkers—such as cholesterol level for heart disease—are substances, processes or characteristics of the body that communicate disease risk, onset and/or progression. They aid in diagnosis and disease management and help researchers stratify for clinical trials and test new drugs quicker by measuring biological changes rather than waiting for clinical improvement. There are no validated biomarkers for Parkinson's disease, a reality researchers are hoping to change with PPMI.
Launched in 2010, PPMI is a longitudinal clinical study that collects standardized clinical, imaging and biologic data. Now taking place at 32 clinical sites around the world, the study completed initial enrollment of 423 recently diagnosed Parkinson's patients and 196 controls in April 2013. That month PPMI began recruiting individuals with the known Parkinson's risk factors of smell loss and REM sleep behavior disorder.
"In the fourth year of PPMI, it is evident that a large-scale biomarker study is not only possible in Parkinson's disease, but is already yielding scientific insights that could help transform the field of Parkinson's research," said Todd Sherer, PhD, CEO of The Michael J. Fox Foundation. "The exceptional investigators at sites around the world, such as Emory University, have created the infrastructure that allows us to make such strides, by working together."