How the body fights against viruses
Scientists of the Max F. Perutz Laboratories of the University of Vienna and the Medical University of Vienna, together with colleagues of the ETH Zurich, have now shown how double stranded RNA, such as viral genetic information, is prevented from entering the nucleus of a cell. During the immune response against viral infection, the protein ADAR1 moves from the cell nucleus into the surrounding cytoplasm. There it modifies viral RNA to inhibit reproduction of the virus. But how is the human genome protected from inadvertent import of viral RNA into the nucleus?
The current study of the research teams from Vienna and Zurich, published in PNAS Plus, sheds the first light on this question.
The human immune system: defense against germs
We are constantly exposed to bacteria and viruses. Luckily our body has developed a set of mechanisms – the immune system – to fend off and fight such attacks. Viruses are small particles that cannot survive outside of a host cell. Upon entering the body they release their genetic material into our cells in order to reproduce. This is one of the points where the body's immune system attacks: enzymes chemically modify the viral genetic information in a manner that renders it useless for the production of new virus particles.
ADAR1 – immunological weapon to fight viruses
ADAR1 is one of the enzymes involved in the antiviral immune response. Usually it is located in the nucleus of the cell, but upon infection, ADAR1 relocates to the cytoplasm. It binds and chemically modifies the virus-RNA so that it cannot be used to produce new virus. But how does the cell make sure that ADAR1 doesn't take the bound viral RNA back into the nucleus? After all, the human genome is stored there and needs to be protected. This question was now investigated by the teams of Michael Jantsch from the Max F. Perutz Laboratories (MFPL) of the University of Vienna and Frédéric Allain from the ETH Zurich. As part of the study, funded by a DACH grant for joint projects of German (D), Austrian (A) and Swiss (CH) research groups, Frédéric Allain and his team solved the structure of the ADAR1 RNA-binding domain. This domain was first identified as an important module for RNA binding and cellular localization by the Viennese team around Michael Jantsch.
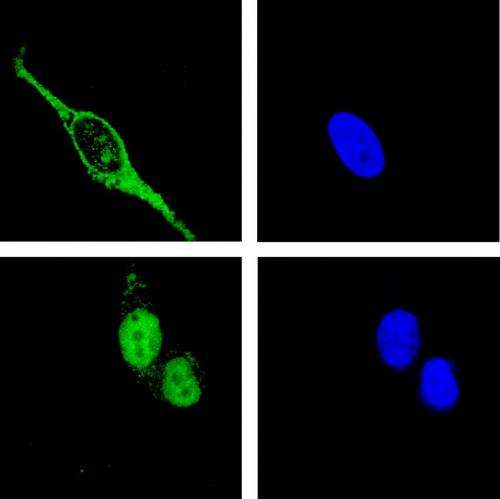
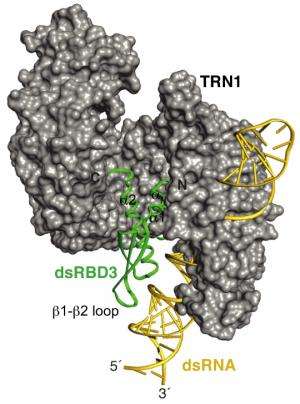
The structure revealed that two modules flanking the RNA-binding region control nuclear transport. Further cell biological analysis by the Viennese team showed that ADAR1 could only relocate into the nucleus when the RNA-binding domain is present to connect the two nuclear transport modules. Michael Jantsch explains: "When we removed the RNA-binding domain, ADAR1 was unable to move into the nucleus. The same was the case when it was bound to viral double-stranded RNA. With the help of the scientists at the ETH we could show how this can be explained structurally."
Which RNAs flip the switch of ADAR1?
The bound RNA actually obstructs the way for ADRA1 to re-enter into the nucleus: the two nuclear transport modules cannot bind their partner that mediates transport across the nuclear envelope because the RNA makes this sterically impossible. "This type of mechanism has never been reported before. You can picture it similarly to driving into a car park. To open the gates you need to get a ticket by pressing a button on the ticket machine. This works fine, unless you transport something really bulky on the roof. In that case the transported goods prevent you from driving close enough to operate the ticket machine", exemplifies Michael Jantsch. Now, he and his team want to identify the RNAs that can actually flip this switch for ADAR1 – so those that can be bound by ADAR1 and prevent its relocation into the nucleus. The researchers are also keen to find out if and which other cellular proteins location are regulated by RNA.
More information: Pierre Barraud, Silpi Banerjee, Weaam I. Mohamed, Michael F. Jantsch and Frédéric H.-T. Allain: A bimodular nuclear localization signal assembled via an extended dsRBD acts as an RNA-sensing signal for Transportin 1. In: PNAS Plus (April 2014). www.pnas.org/cgi/doi/10.1073/pnas.1323698111