'3-D' test could reduce reliance on animals for testing asthma and allergy medications
To determine whether new medicines are safe and effective for humans, researchers must first test them in animals, which is costly and time-consuming, as well as ethically challenging. In a study published in ACS' journal Molecular Pharmaceutics, scientists report that they've developed a simple, "3D" laboratory method to test asthma and allergy medications that mimics what happens in the body, which could help reduce the need for animal testing.
Amir Ghaemmaghami and colleagues note that respiratory conditions, such as asthma and allergies, are becoming more common. These conditions affect the lungs and the airway leading to the lungs, making it difficult to breathe. Every year, respiratory symptoms lead to expensive hospital visits, as well as absences from work and school. Better drugs could provide relief, but before giving new medicines to people, researchers must first test them in animals—a costly and laborious process. Sometimes, researchers will use "2D" tests in which they apply the drug to a layer of human cells in a lab dish instead, but this isn't an adequate way to tell how a medicine will work in a whole animal or a whole person. So, Ghaemmaghami's team developed a new, 3D alternative.
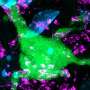
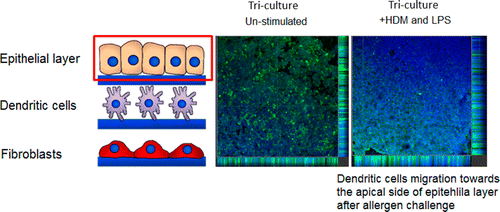
Their test includes three types of human cells that are typically in a person's airway. In the body, these cells are close together and are involved in the development of respiratory conditions. The 3D "model" reacted just like a real person's airway when they exposed it to allergens and bacterial extract. They say that the model has the potential of reducing the need for some animal testing of new drugs for respiratory conditions.
More information: "Immunocompetent 3D Model of Human Upper Airway for Disease Modeling and In Vitro Drug Evaluation" Mol. Pharmaceutics, Article ASAP. DOI: 10.1021/mp5000295
Abstract
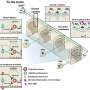
The development of more complex in vitro models for the assessment of novel drugs and chemicals is needed because of the limited biological relevance of animal models to humans as well as ethical considerations. Although some human-cell-based assays exist, they are usually 2D, consist of single cell type, and have limited cellular and functional representation of the native tissue. In this study, we have used biomimetic porous electrospun scaffolds to develop an immunocompetent 3D model of the human respiratory tract comprised of three key cell types present in upper airway epithelium. The three cell types, namely, epithelial cells (providing a physical barrier), fibroblasts (extracellular matrix production), and dendritic cells (immune sensing), were initially grown on individual scaffolds and then assembled into the 3D multicell tissue model. The epithelial layer was cultured at the air–liquid interface for up to four weeks, leading to formation of a functional barrier as evidenced by an increase in transepithelial electrical resistance (TEER) and tight junction formation. The response of epithelial cells to allergen exposure was monitored by quantifying changes in TEER readings and by assessment of cellular tight junctions using immunostaining. It was found that epithelial cells cocultured with fibroblasts formed a functional epithelial barrier at a quicker rate than single cultures of epithelial cells and that the recovery from allergen exposure was also more rapid. Also, our data show that dendritic cells within this model remain viable and responsive to external stimulation as evidenced by their migration within the 3D construct in response to allergen challenge. This model provides an easy to assemble and physiologically relevant 3D model of human airway epithelium that can be used for studies aiming at better understanding lung biology, the cross-talk between immune cells, and airborne allergens and pathogens as well as drug delivery.