Toward a faster, more accurate way to diagnose stroke
When someone suffers from a stroke, a silent countdown begins. A fast diagnosis and treatment can mean the difference between life and death. So scientists are working on a new blood test that one day could rapidly confirm whether someone is having a stroke and what kind. Their report appears in the ACS journal Analytical Chemistry.
Steven A. Soper and colleagues note that strokes, which are the third leading cause of death and disability in the United States, have two possible causes. In ischemic strokes, a clot stops blood flow in a part of the brain. In hemorrhagic stroke, a blood vessel in the brain ruptures. Both can lead to similar symptoms, such as numbness on one side of the body, sudden weakness and headache. Current diagnostic tests can't tell between these two types. But treatment, ideally within three hours of onset, depends on the kind of stroke a person is having. Soper's collaborator, Alison Baird, who is at SUNY Downstate Stroke Center, found clues—or biomarkers—in the blood that can suggest the stroke type and assist in determining the course of proper treatment. Soper's team sought a way to detect those clues quickly.
They built a device that can process whole blood and isolate genetic material for two potential stroke biomarkers within minutes. Keeping in mind that identifying more biomarkers could aid in diagnosis, they designed their device so it can analyze a total of four biomarkers at the same time.
More information: "Parallel Affinity-Based Isolation of Leukocyte Subsets Using Microfluidics: Application for Stroke Diagnosis" Anal. Chem., Article ASAP. DOI: 10.1021/ac5007766
Abstract
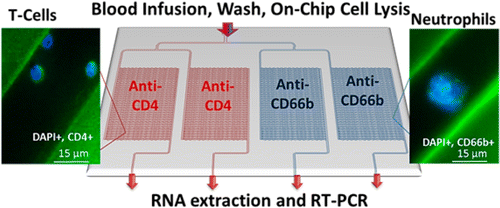
We report the design and performance of a polymer microfluidic device that can affinity select multiple types of biological cells simultaneously with sufficient recovery and purity to allow for the expression profiling of mRNA isolated from these cells. The microfluidic device consisted of four independent selection beds with curvilinear channels that were 25 μm wide and 80 μm deep and were modified with antibodies targeting antigens specifically expressed by two different cell types. Bifurcated and Z-configured device geometries were evaluated for cell selection. As an example of the performance of these devices, CD4+ T-cells and neutrophils were selected from whole blood as these cells are known to express genes found in stroke-related expression profiles that can be used for the diagnosis of this disease. CD4+ T-cells and neutrophils were simultaneously isolated with purities >90% using affinity-based capture in cyclic olefin copolymer (COC) devices with a processing time of 3 min. In addition, sufficient quantities of the cells could be recovered from a 50 μL whole blood input to allow for reverse transcription-polymerase chain reaction (RT-PCR) following cell lysis. The expression of genes from isolated T-cells and neutrophils, such as S100A9, TCRB, and FPR1, was evaluated using RT-PCR. The modification and isolation procedures demonstrated here can also be used to analyze other cell types as well where multiple subsets must be interrogated.















