Researchers pursuing powdered nasal vaccine against West Nile
Riding the expansion of mosquito-borne diseases from the tropics as temperatures rise across the globe, the West Nile Virus made its American landfall in New York about 15 years ago. Since then, it has spread throughout the United States and Canada, killing more than 1,600 and sickening nearly 40,000, according to the Centers for Disease Control and Prevention.
There isn't any human vaccine to control outbreaks of West Nile Virus or to stem its worldwide spread.
But Herman Staats, Ph.D., and Soman Abraham, Ph.D, both professors of pathology at Duke University, are trying to change that. They are combining their expertise in vaccine development and immune responses toward the goal of designing "needleless" vaccines—nasal sprays like the seasonal Flumist vaccine for influenza – that can protect the public against emerging infectious diseases like anthrax or West Nile Virus.
"When vaccinating for an outbreak, the goal is to give the vaccine to as many people as possible, as quickly as possible," says Staats. "But traditional vaccines won't fit the bill, because they call for trained personnel to administer the injection, require three doses to achieve protection, and have a limited shelf life."
From the outset, Staats and Abraham sought to overcome these three shortcomings of vaccines.
Their initial task was to figure out a way to vaccinate against the West Nile Virus that wouldn't require a needle stick and would instead target the mucus-covered surfaces inside the nose. They knew that for such an approach to be successful, the vaccine would need to contain a few key ingredients.
The main ingredient, as with all vaccines, would be a portion of the offending virus called an antigen. Just as a piece of clothing can put dogs on a trail to track down a criminal, an antigen can arouse cells of the immune system to go after a virus once it has invaded the body. In this case, they included a fragment of the outer surface of the virus called "West Nile Virus envelope domain 3" in the vaccine formulation.
To this central component, the researchers next needed to add a special ingredient known as an adjuvant, a substance that amps up the immune response to make the vaccine even more powerful. Over the years, Staats has tested a slew of adjuvant candidates – from toxic agents like mutant cholera toxins to intracellular messengers known as cytokines. In 2008, Staats and Abraham happened upon a particularly promising family of molecules – known as mast cell activators—that could enhance the potency of vaccines with little or no side effects.
"Most people think of mast cells as the cell type responsible for allergic and anaphylactic reactions, but they also play a protective role in fighting microbial infection," says Abraham. "By adding small peptides or synthetic compounds that activate mast cells to our vaccines, we can simulate what would happen in the course of a natural infection. The activated mast cells will release tiny granules that contain cytokines and other immune activators, and those will recruit other immune cells, enhancing the immune response to the vaccine antigen."
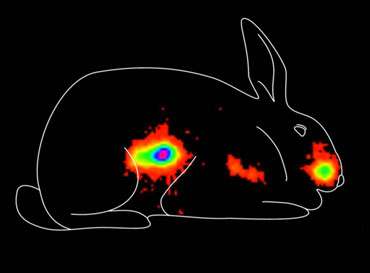
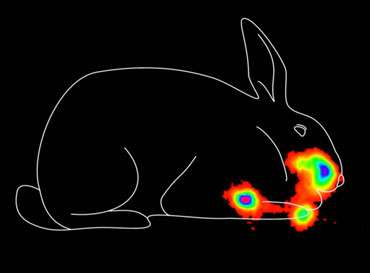
WNV vaccineAfter 3 hours, much of the liquid vaccine is still in the nose, but most has also moved to the gut, where it won't do any good.
To this cocktail, they added one final ingredient – another adjuvant to boost the potency of the vaccine even further. While the first adjuvant activates the mast cells, this one – a strip of bacterial DNA – stimulates the cells that present the vaccine's main antigenic ingredient to the immune system.
With these three tools—the two adjuvants and the antigen—a vaccine can be sprayed into the nose to mobilize the immune arsenal. These infection-fighters then migrate from the nasal cavity into nearby lymph nodes before circulating throughout the entire bloodstream to protect against a future attack.
Less is more
Because most vaccinations of this type – such as hepatitis B or HPV vaccines – are delivered in three doses, Staats looked for a dosing schedule that could induce immunity to the West Nile Virus after three rounds. Working in mice, he discovered that three doses of 15 micrograms of antigen – a tiny amount – given over the course of three weeks could provide partial protection against the disease.
But in an actual outbreak, three weeks would be too long to wait for protection. The researchers began to wonder if it was possible to do the trick in just two doses.
By upping the amount of antigen from 15 to 45 micrograms, the researchers were able to achieve the same result in just two doses over two weeks. What's more, they found that increasing the amount to 60 micrograms gave full protection after two doses against the West Nile Virus.
Though promising, the results achieved in mice are not always easily translated to humans. Mice have much smaller noses than humans, which could affect the delivery and behavior of mucosal vaccines.
Therefore, Staats has been repeating a number of experiments in rabbits, which have nasal cavities comparable in size to humans. But he found that when he sprayed the vaccine into the larger nasal cavity of the rabbits, a portion of the liquid was swallowed or dripped out of the nose before it could do its job.
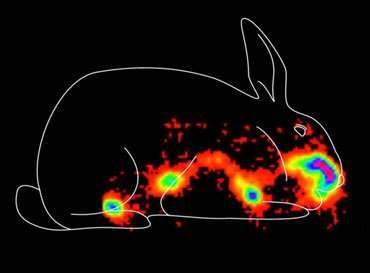
So they moved on to develop a dry powder version of the vaccine. For this step, the researchers brought in the expertise of the University of Oklahoma's Dr. Lucila Garcia-Contreras and RTI International's Dr. Anthony Hickey.
Just as with the liquid preparation, the powder can be sprayed into the nasal cavity. But instead of being swallowed, the powder adheres to the moisture in the nasal passages, immediately reconstituting the vaccine so it can get to work. Not only are dry vaccines easier to deliver, but they are also easier to stockpile. Powdered vaccine requires no refrigeration and has been found to remain potent even after two and a half years of storage.
They presented their latest findings at the American Society for Microbiology's 2014 Biodefense and Emerging Diseases meeting.
The team is currently testing the dry powder, two-dose, nasal vaccine in rabbits. Once they work out all the kinks, they plan to continue perfecting the West Nile Virus vaccine for human use and also to begin exploring the application of this technology to other vaccines.
"Although we are working on West Nile Virus right now, what we learn from this vaccine system could also be applied to the yearly flu vaccine or the tetanus booster," says Staats. "Our ultimate goal is to develop adjuvants, formulations, and delivery methods that can be suitable for any vaccine."