Scientists cast light on the brain's social cells
Picture yourself hovering over an alien city with billions of blinking lights of thousands of types, with the task of figuring out which ones are connected, which way the electricity flows and how that translates into nightlife. Welcome to the deep brain.
Even in an era rapidly becoming known as the heyday of neuroscience, tracing the biochemical signaling among billions of neurons deep in the brain has remained elusive and baffling.
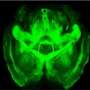
A team of Stanford University researchers managed to map out one such connection, buried inside the brain of a living, moving mammal as they manipulated its behavior. The feat offers an unprecedented close-up of the genesis of social behavior on a cellular level, and could offer insights into psychiatric puzzles such as autism, depression and anxiety.
"It's a new kind of data that no one has been able to get before - a single kind of cell projecting from one deep brain area to another deep brain area during behavior," said Stanford bioengineer and neuroscientist Karl Deisseroth, senior author of the study published online this week in the journal Cell.
Deisseroth's team relied on genetics, fiber-optics and a bunch of female mice.
The Deisseroth lab at Stanford had already pioneered the use of optogenetics in neuroscience, a technique that delivers light through a hair-thin probe to stimulate cells that have been modified with a light-sensitive gene. First demonstrated in 2007, optogenetic stimulation not only changed the scale and precision involved in exploring the brain, it allowed researchers to better discern cause and effect, which often were muddled by conventional imaging and detection devices.
Researchers soon began using the technique widely to manipulate the brain cells of laboratory animals. They discovered that stimulating one brain cell had a profound effect on behavior.
Finding out how this works, however, depended on tracing the connections, or "projections," from the stimulated neurons.
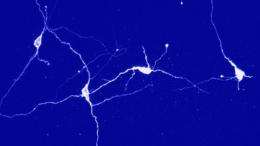
The deep brain is a very "noisy" place. The tiny voltage changes that propagate along axons, the slender fibers that extend from the nucleus of a neuron, are difficult to distinguish. Researchers routinely add fluorescent properties to the calcium ions that help drive these voltage change in axons, so they can "see" large-scale evidence of activity. But no one had been able to track that signal in an axon while an animal reacted to the stimulation.
"It's buried in the noise and it's too small to see in a behaving animal," Deisseroth said. "We've never been able to see it. We've never been able to observe how animals normally use projections."
The Stanford team tried a new trick. The researchers delivered the light at a specific frequency by chopping it up with what amounts to a fancy pinwheel. Since the calcium ions fluoresce at the same frequency as the incoming light, the team designed a device to pick up only that signal. That allowed them to follow the signal in real time while they chronicled the animal's behavior. They call the new technique fiber photometry.
The rest was relatively simple rodent play. Lab members placed the probes in the areas they had altered for optogenetic stimulation, set up the detection instruments, then ran trials to test the mouse's reaction to other mice.
©2014 Los Angeles Times
Distributed by MCT Information Services