Cooling treatment improves survival without brain damage in later childhood
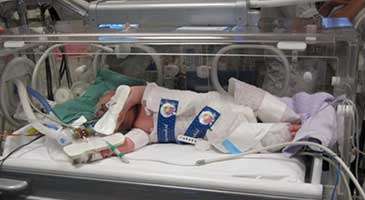
(Medical Xpress)—The cooling of newborn babies suffering from perinatal asphyxia – a lack of oxygen at the time of birth – significantly increases their chance of survival without brain damage to later childhood (age six to seven years), according to a Medical Research Council (MRC) funded clinical trial by a team of researchers from the universities of Bristol, Oxford, Leeds, University College London, Imperial College London, Queen's University Belfast and Homerton University Hospital, London.
The study, published in the New England Journal of Medicine, found that 51.7 per cent of infants treated with hypothermia survived with an IQ of 85 or above, which is considered to be within the normal range, compared to 39.4 per cent of those treated with standard care. Cooling significantly reduced the chance of oxygen-deprived children suffering from cerebral palsy and other moderate/severe disabilities. The children also showed improved motor functioning. However, the authors observed that there was no difference in mortality rate – reported to be around 30 per cent of children enrolled in the trial – between the standard care and hypothermia-treated groups.
The trial, led jointly by the National Perinatal Epidemiology Unit (NPEU) at the University of Oxford and Imperial College London, and with Professor Marianne Thoresen and Professor Andrew Whitelaw from the University of Bristol's School of Clinical Sciences, is the largest study of its kind and the first study to show improved brain function in children treated using this method in later life. Before this trial, there was limited information on the beneficial effect of cooling after asphyxia beyond the age of 18 months. This work is important because it demonstrates that the improvements observed in brain function are not just temporary.
The MRC TOBY trial (TOBY: Total Body Hypothermia) involved newborn babies of at least 36 weeks gestation that suffered from a lack of oxygen at birth. The children were randomly assigned into two groups within six hours of delivery, and either treated with standard care or standard care plus hypothermia, when their body temperature was reduced to 33.5˚C for 72 hours. After that time, they were slowly returned to a normal body temperature of 37˚C.
The current research is part of the follow up TOBY Children Study that aimed to find out if there were any differences in the health of the children, treated with or without cooling, in later life. The scientists tested the children's mental abilities and performance at school, looked at parent and teacher reports on behaviour and investigated the presence and severity of any disabilities that resulted from oxygen deprivation.
Following oxygen deprivation a number of processes are set off in the brain, which lead to brain cell death and permanent neurological damage. Hypothermia interrupts these processes to reduce brain injury and has consistently been shown to improve outcomes at 18 months. This study now confirms treatment with cooling is safe and effective, and that the benefits persist in the long term. The treatment has been endorsed by the National Institute for Health and Care Excellence (NICE) and adopted into clinical practice in the NHS; it has the advantage of being relatively simple and inexpensive to carry out. These new findings support its general use in neonatal clinical practice.
Professor Denis Azzopardi, of King's College London and lead author of the study, said: "This study is important as it confirms improved brain function persisting into middle childhood with cooling treatment and it is a proof of the concept that treatment following oxygen deprivation at birth can be effective."
Professors Marianne Thoresen and Andrew Whitelaw, who were involved in the design, recruitment, data collection and analysis of the MRC TOBY trial, added: "After 20 years of research starting in the lab and then progressing to clinical trials, we now have good evidence that cooling after severe lack of oxygen can protect babies' brains with lasting effect.
"Bristol played a key role in the early experimental work and in this trial which showed that cooling for 72 hours improves the chances of surviving with normal function at school age. It is important that hypoxic brain injury is recognised as an emergency after birth as cooling must be started as soon as possible. However, many babies are still disabled every year and there is a need to develop new treatments which add to the brain protection from cooling."
Brenda Strohm, research nurse and TOBY trial coordinator at the NPEU, University of Oxford, said: "We are indebted to all the families who took part in the TOBY Trial and then the TOBY Children Study; their contribution to these two important studies has made a real difference to neonatal care."
"While corresponding with the families over the years, I have shared many happy stories of achievement and success as well as moments of sadness and loss; this is a privilege which adds a special dimension to my role of Study Co-ordinator. Thanks to our latest research, now when a baby is treated with cooling, families can be more fully informed about what might lie ahead, not only in the coming months but in the next few years too."
Hugh Perry, chair of the MRC Neurosciences and Mental Health Board, said: "This study is a great example of how research can change people's lives. Although major advances have been made in how childbirth is managed, approximately two out of every 1,000 newborn infants suffer from a lack of oxygen around the time of birth. Prior to the introduction of cooling therapy there were no approved, specific treatments that reduced the risk of brain injury long-term following asphyxia."
More information: "Effects of Hypothermia for Perinatal Asphyxia on Childhood Outcomes." Denis Azzopardi, M.D., Brenda Strohm, R.N., Neil Marlow, D.M., Peter Brocklehurst, F.F.P.H., Aniko Deierl, M.D., Ph.D., Oya Eddama, Ph.D., Julia Goodwin, Ph.D., Henry L. Halliday, M.D., Edmund Juszczak, M.Sc., Olga Kapellou, M.D., Malcolm Levene, M.D., Louise Linsell, M.Sc., Omar Omar, M.Sc., Marianne Thoresen, M.D., Ph.D., Nora Tusor, M.D., Andrew Whitelaw, M.D., and A. David Edwards, D.Sc. for the TOBY Study Group. N Engl J Med 2014; 371:140-149. July 10, 2014. DOI: 10.1056/NEJMoa1315788