July 9, 2014 report
Microglia: the movers and shakers of the brain
(Medical Xpress)—Once the brain has fully developed, its neurons and astrocytes remain relatively fixed. There are a few select migratory streams that replenish vulnerable populations with dedicated stem cell recruits, but by and large the status quo holds sway. The microglial cell (MG) is an exceptional creature that doesn't play by these rules. Born in the foreign lands of the early embryo's yolk sac, MGs migrate into the developing neural tube through the primitive bloodstream and never quite settle down. Just what it is these special license wild rovers get up to in the brain is the subject of a comprehensive new review in the Journal Cell.
Authors Michael Salter and Simon Beggs from the University of Toronto make it clear in their paper that MGs do much more than just create inflammation in response to invaders. More often then not, their awesome powers of mobility are turned inward to police the brain's own cells. While calling a synapse "inappropriate" makes about as much sense as calling a child illegimate, some synapses would appear to be a little too much. It is now well established that MGs play an essential role in cutting back not just synapses, but whole branches of neurites. The question is not just how they do they do this, but also how do they know they should do it?
Perhaps the best way to understand how the "immune cells" of the brain work is to take a look at their kindred spirits in other organ systems. As an immuno-privileged organ, many of mechanisms used in the body to fight invaders don't need to operate to full capacity in the brain. However that's not to say they are absent. The tireless chronicler of individual cell intelligence Jon Lief observes there are actually many immune cells that make regular transits through the cerebral spinal fluid including B and T lymphocytes, macrophages, mast cells, and dendritic cells.
We usually think of neurons as the "smart" cells of the body. Yet compared to the macrophages or T cells which patrol the entire body, neurons don't outwardly appear to do all that much. Within each organ, there is a resident population of macrophages that take on a unique form suited to the local needs: The hostile environment of the alveolar macrophage, for example, places huge phagocytic demands on the cell as it maintians the border between body and air. The osteoclasts of bone are the mechanical engineers of the body who sense macroscopic forces from worlds beyond and sculpt the calcium clay indiscriminately slopped by osteocytes that have entombed themselves within their invidiual caniculi.
Likewise, the liver has its own unique Kuppfer cells, the placenta its Hofbauer cell, and so on till your hearts content. Calling MG cells the macrophages of the brain would imply to some that all they do is clean up. However as once-opaque developmental mysteries continue to give up their secrets we see that macrophage-like cells are more like commanders than pawns. These great revisionists do everything from regenerate the of limbs of Salamandars to melt the webbing between finger and toes.
Perhaps the most definitive, and intriguing role of MG is their critical part in the segregation of inputs from the eyes within the visual system. Classical compliment cascade proteins (like C1q and C3) are repurposed from the immune system to "opsonize" (from Greek, prepare for eating) or tag exuberant synapses for elimination. In the vertebrate immune system these innate proteins help or "compliment" the more adaptive T cell antibody systems. To further expand the scope of what is possible in all things cellular, we might consider here some of the more rarefied superpowers that T cells posses.
Like MGs, T cells are in constant communication with more social partners than a teenager on Instagram. Yet through all this the persist like a lone wolf, seeming to act primarily for numero uno as they administer their own territory. Rather then needing to idle away as neurons do in endless volley of spike trains, T cells can make rapid metabolic adjustments for bursts of activity when needed. They are also able to continually optimize antibody interactions through hypermutation. This ability to self edit their own DNA to make diversify receptors through in-situ recombination is virtually unparalleled elsewhere in nature. The closest thing to it we might imagine for the brain might be the tiling of the full unknowable receptor space in the olfactory system. Only here every single odorant receptor gets translating from its own specific gene.
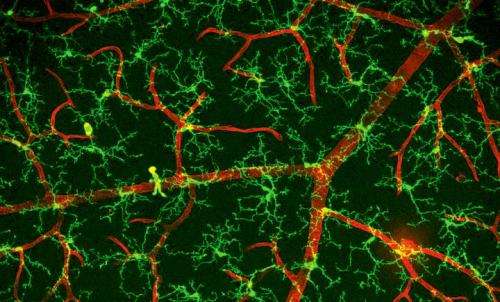
We know from the anatomical and molecular studies of the visual system, that once in, MGs get their hands into everything. In the LGN, the part of the thalamus where inputs from the retina round, processess from MG cells coat both pre and post synaptic elements. If things here want to move, it seems they need to go through the MGs. For the incoming optic nerve, this is all part of the circle of life as peripheral myelinating Schwann cells yeild more centrally to the CNS-ensheathing oligodendroctyes, which yeild ultimately to the synaptic MG engulfers. That is not to say that MG are entirely synapse-oriented—their initial discoverer and namer, Pío del Río Hortega, found them spanning the corpus callosum.
For us, the most important general concern may be exchanges between brain and body. The migration of MG into the brain (and for that matter perhaps out as well) is shut down when the blood brain barrier forms early in development. None-the-less immune cells, stem cells, and just about anything else can find its way in when things get compromised. Stories of mom's with Y chromsomes in their brains from the boys they previously carried, or stem cells injected into the blood that fuse to the nearest partner once in the brain abound. These things are not mere anomalies in the potluck of genetic recombination but are rather par for the course of life.
The review paper mentions the recent discovery and classification of sub-populations of Mg-like cells. Extensive transcriptome analysis of all the proteins expressed by these cells has provided a rigorous way, at least in theory, to catagorize them as they pertain to diseases. Hoxb8-expressing cells, which represent about 15% of the CD11b+ MG cells in general population are believed to be ontogenically distinct from the yolk cell population. That suggests that they might come in later from elsewhere. Understanding what controls the ability of these cells to gain access to the brain, and move about it will be critical to further understanding their function.
More information: Sublime Microglia: Expanding Roles for the Guardians of the CNS, Cell, Volume 158, Issue 1, p15–24, 3 July 2014. www.cell.com/cell/abstract/S0092-8674(14)00729-6
Abstract
Recent findings challenge the concept that microglia solely function in disease states in the central nervous system (CNS). Rather than simply reacting to CNS injury, infection, or pathology, emerging lines of evidence indicate that microglia sculpt the structure of the CNS, refine neuronal circuitry and network connectivity, and contribute to plasticity. These physiological functions of microglia in the normal CNS begin during development and persist into maturity. Here, we develop a conceptual framework for functions of microglia beyond neuroinflammation and discuss the rich repertoire of signaling and communication motifs in microglia that are critical both in pathology and for the normal physiology of the CNS.
© 2014 Medical Xpress