Exercising restraint to stall tumor growth
Many proteins undergo an assembly line-style process of glycosylation as they travel from a cellular structure called the endoplasmic reticulum (ER) to the Golgi apparatus and on through its various compartments, after which they are released. Disruptions in this process can contribute to a variety of diseases. Now researchers from A*STAR have identified a regulatory mechanism that prevents the production of glycosylated proteins that potentially promote cancerous growth1.
The glycosylation process—executed by various enzymes in a stepwise fashion—attaches complex sugar molecules onto proteins, which can fundamentally alter the destination and function of these proteins in the cell. Many tumors produce proteins that are known as 'Tn antigens': these proteins have been decorated with a single sugar molecule by enzymes from the N-Acetylgalactosaminyltransferase (GalNAc-T) family.
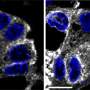
GalNAc-T enzymes normally reside within the Golgi, but Frederic Bard and colleagues at the A*STAR Institute of Molecular and Cell Biology in Singapore previously showed that these enzymes relocate to the ER in certain tumors2. Here they target resident proteins for modification and thus contribute to Tn production.
To better understand why this happens, Bard's team screened more than 900 regulatory proteins that affect Tn levels. Remarkably, their experiments uncovered a dozen different proteins that appear to ensure that GalNAc-T enzymes are retained within the Golgi instead of being delivered to the ER. "We were surprised by the number of regulators that we could identify," says Bard. One of these, a signaling protein called extracellular signal regulated kinase 8 (ERK8), appeared to be particularly important for restricting Tn accumulation to the Golgi.
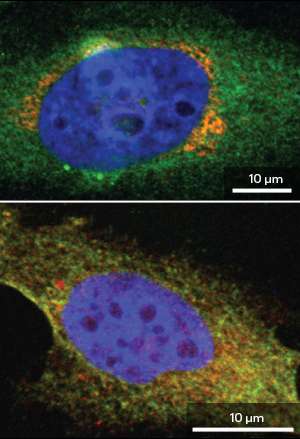
When the researchers inhibited production of ERK8, they observed a clear redistribution of GalNAc-Ts from the Golgi to the ER, indicating that this protein normally puts the brakes on such 'backward' traffic (see image). "After knocking down ERK8, the relocation pathway responds like a compressed spring that has just been released," says Bard. The subsequent production of Tn antigens stimulates the active cell migration observed in aggressive cancers, and ERK8 similarly appears to act as a critical check against this behavior in cultured cancer cells.
The researchers also noted unusually low ERK8 levels in tumor tissue taken from breast and lung cancer patients. Bard's team is now working with animal models to determine the importance of this protein as a safeguard against cancerous growth. "We are building a mouse model where we can experimentally remove ERK8 and observe the effects in healthy and cancer tissues," says Bard. "We predict that loss of ERK8 will promote tumor invasiveness."
More information: 1. Chia, J., Tham, K. M., Gill, D. J., Bard-Chapeau, E. A. & Bard, F. A. ERK8 is a negative regulator of O-GalNAc glycosylation and cell migration. eLife 3, e01828 (2014). dx.doi.org/10.7554/eLife.01828
2. Gill, D. J., Tham, K. M., Chia, J., Wang, S. C., Steentoft, C. et al. Initiation of GalNAc-type O-glycosylation in the endoplasmic reticulum promotes cancer cell invasiveness. Proceedings of the National Academy of Sciences USA 110, E3152–E3161 (2013). dx.doi.org/10.1073/pnas.1305269110