Potential new treatment strategy for aggressive leukemia
Scientists have found a potential treatment strategy for an aggressive type of leukemia by targeting enzymes used by cells to sense and adapt to oxygen levels.
Apr 18, 2024
0
37
Scientists have found a potential treatment strategy for an aggressive type of leukemia by targeting enzymes used by cells to sense and adapt to oxygen levels.
Apr 18, 2024
0
37

Infants and children who have severe cases of respiratory syncytial virus (RSV) before age 2 are likely to have changes to their lung structure and function that could affect respiratory health later in life.
Apr 18, 2024
0
0

White cells called regulatory B cells could be harnessed to fight cancers and to treat autoimmune diseases by either increasing or decreasing their function, as indicated by a new research.
Apr 17, 2024
0
8

Hypoxia-activated prodrugs (HAPs) have the potential to activate specifically in hypoxic tumors and eliminate tumor cells, which has brought new opportunities for safe and effective anticancer therapy. However, their therapeutic ...
Apr 3, 2024
0
0

The brain consumes approximately 25% of the body's oxygen to fuel its neural activities, underscoring the importance of sufficient oxygen supply for maintaining normal cognitive operations. Consequently, monitoring cerebral ...
Apr 2, 2024
0
41

Aerobic exercise like jogging, biking, swimming or hiking is a fundamental way to maintain cardiovascular and overall health. The intensity of aerobic exercise is important to determine how much time you should spend training ...
Apr 2, 2024
0
0

The human brain consumes vast amounts of energy, which is almost exclusively generated from a form of metabolism that requires oxygen. While the efficient and timely delivery of oxygen is known to be critical to healthy brain ...
Mar 28, 2024
0
30
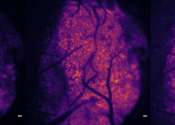
University of Calgary researchers have discovered the lungs communicate directly with the brain when there is an infection. Findings show that the brain plays a critical role in triggering the symptoms of sickness, which ...
Mar 21, 2024
0
36

Physicians and biomedical engineers at Duke University have developed a method to visualize the growth of a placenta throughout a mouse's pregnancy. By coupling an implantable window with ultrafast imaging tools, the approach ...
Mar 21, 2024
0
13
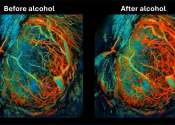
Australian researchers have discovered that a bedtime nasal spray has the potential to reduce the severity of sleep apnea in people and lower their blood pressure.
Mar 15, 2024
0
68

Oxygen (pronounced /ˈɒksɨdʒɨn/, from the Greek roots ὀξύς (oxys) (acid, literally "sharp", from the taste of acids) and -γενής (-genēs) (producer, literally begetter) is the element with atomic number 8 and represented by the symbol O. It is a member of the chalcogen group on the periodic table, and is a highly reactive nonmetallic period 2 element that readily forms compounds (notably oxides) with almost all other elements. At standard temperature and pressure two atoms of the element bind to form dioxygen, a colorless, odorless, tasteless diatomic gas with the formula O2. Oxygen is the third most abundant element in the universe by mass after hydrogen and helium and the most abundant element by mass in the Earth's crust. Diatomic oxygen gas constitutes 20.9% of the volume of air.
All major classes of structural molecules in living organisms, such as proteins, carbohydrates, and fats, contain oxygen, as do the major inorganic compounds that comprise animal shells, teeth, and bone. Oxygen in the form of O2 is produced from water by cyanobacteria, algae and plants during photosynthesis and is used in cellular respiration for all complex life. Oxygen is toxic to obligately anaerobic organisms, which were the dominant form of early life on Earth until O2 began to accumulate in the atmosphere 2.5 billion years ago. Another form (allotrope) of oxygen, ozone (O3), helps protect the biosphere from ultraviolet radiation with the high-altitude ozone layer, but is a pollutant near the surface where it is a by-product of smog. At even higher low earth orbit altitudes monatomic oxygen (O1) is a significant presence and a cause of erosion for spacecraft.
Oxygen was independently discovered by Carl Wilhelm Scheele, in Uppsala, in 1773 or earlier, and Joseph Priestley in Wiltshire, in 1774, but Priestley is often given priority because his publication came out in print first. The name oxygen was coined in 1777 by Antoine Lavoisier, whose experiments with oxygen helped to discredit the then-popular phlogiston theory of combustion and corrosion. Oxygen is produced industrially by fractional distillation of liquefied air, use of zeolites to remove carbon dioxide and nitrogen from air, electrolysis of water and other means. Uses of oxygen include the production of steel, plastics and textiles; rocket propellant; oxygen therapy; and life support in aircraft, submarines, spaceflight and diving.
This text uses material from Wikipedia, licensed under CC BY-SA