Chopping off protein puts immune cells into high gear
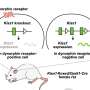
The complex task of launching a well-organized, effective immune system attack on specific targets is thrown into high gear when either of two specific enzymes chop a protein called LAG-3 off the immune cells leading that battle, according to investigators at St. Jude Children's Research Hospital.
These cells, called T lymphocytes, are key to the body’s ability to fight off infections, tailoring the immune response so it focuses on specific targets. When activated, certain T lymphocytes called effector T cells reproduce, increasing their numbers and enhancing their ability to protect the body.
The St. Jude finding is important because it represents a new concept in how T cells are regulated, according to Dario Vignali, Ph.D., associate member of the St. Jude Department of Immunology. The study offers the first example of a protein that is required for dampening T cell activity being controlled by getting chopped off at the T cell’s surface. Certain drugs that inhibit metalloproteases now under development as treatments for multiple sclerosis and arthritis appear to work by keeping T cells on a tight leash, Vignali noted. The new discovery could demonstrate an additional way in which these drugs work. Vignali is senior author of a report on this work that appears in the January 24 issue of The EMBO Journal.
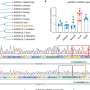
The investigators performed their studies using animal cells that were genetically modified to carry LAG-3 on their surface; the researchers also used drugs that inhibit enzymes that chop off LAG-3. The team demonstrated that the two enzymes that cleave LAG-3 are controlled by of distinct but overlapping signals generated from the T cell receptor, a specialized protein that allows T lymphocytes to “see” the outside world. The investigators showed that the T cell receptor generates a different, specific signal to control the activity of these metalloprotease enzymes, called ADAM10 and ADAM17.
Specifically, the team demonstrated that ADAM10 normally cleaves LAG-3 even before the T cells are activated. After the T cell receptor receives signals from the immune system, it causes the gene for ADAM10 to make much more of this enzyme, substantially increasing the rate of LAG-3 cleavage. However, ADAM17 is inactive until the T cell receptor triggers a molecule called protein kinase C theta to activate this enzyme. In either case, when metalloproteases remove LAG-3, the brakes are taken off T cell activity.
“Appropriate control of T cell expansion during an immune response is critical,” Vignali said. “We have uncovered a new paradigm in which specialized cell surface enzymes control this process by modulating the expression of a molecule, LAG-3, that acts as an immunological molecular brake. In turn, this process is controlled by the strength of the T cell receptor signal—the immunological ‘accelerator.’ So the more the T cell ‘accelerates,’ the more the ‘brake’ is released.”
The St. Jude team previously reported that regulatory T cells, which prevent effector T cells from running out of control and causing damage to the body, use LAG-3 to rein in these activated effector T cells. The current study in EMBO extends that finding by showing that cleavage of LAG-3 proteins on the surface of T cells allows them to greatly increase their proliferation rate during such a battle. The team also showed that cleaved pieces of LAG-3 do not contribute to T cell control, but are rather “waste” products that are swept away later.
Source: St. Jude Children's Research Hospital