A new study has identified what may be a pivotal first step towards the regeneration of nerve cells following spinal cord injury, using the body’s own stem cells.
This seminal study, published in this week’s Proceedings of the National Academy of Science, identifies key elements in the body’s reaction to spinal injury, critical information that could lead to novel therapies for repairing previously irreversible nerve damage in the injured spinal cord.
Very little is known about why, unlike a wound to the skin for example, the adult nervous system is unable to repair itself following spinal injury. This is in contrast to the developing brain and non-mammals which can repair and regenerate after severe injuries. One clue from these systems has been the role of stem cells and their potential to develop into different cell types.
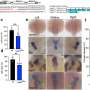
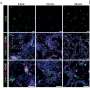
“Because of their regenerative role, it is crucial to understand the movements of stem cells following brain or spinal cord injury,” says Dr. Philip Horner, co-lead investigator and neuroscientist at the University of Washington. “We know that stem cells are present within the spinal cord, but it was not known why they could not function to repair the damage. Surprisingly, we discovered that they actually migrate away from the lesion and the question became why – what signal is telling the stem cells to move.”
The researchers then tested numerous proteins and identified netrin-1 as the key molecule responsible for this migratory pattern of stem cells following injury. In the developing nervous system, netrin-1 acts as a repulsive or attractive signal, guiding nerve cells to their proper targets. In the adult spinal cord, the researchers found that netrin-1 specifically repels stem cells away from the injury site, thereby preventing stem cells from replenishing nerve cells.
“When we block netrin-1 function, the adult stem cells remain at the injury site,” says Dr. Tim Kennedy, co-lead investigator and neuroscientist at the Montreal Neurological Institute of McGill University. “This is a critical first step towards understanding the molecular events needed to repair the injured spinal cord and provides us with new targets for potential therapies.”
Source: Montreal Neurological Institute and Hospital