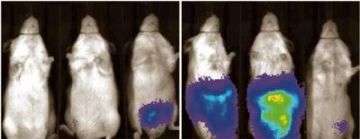
The image shows mice which received similar injections but which had been treated with the anticancer drug PS-341 (on the left), and with zoledronic acid (on the right). Credit: Photos courtesy of Thomas Rosol, Ohio State University
COLUMBUS, Ohio – Researchers here have figured out a way to use a firefly gene to let them see just how effective a new drug combination actually is against some forms of cancer and its serious complication.
The new study looked at ATLL, adult T-cell lymphoma and leukemia, a form of cancer where it is particularly hard to gauge the disease's progress, and where the patients' prognosis is generally poor. There is now no widely effective therapy available to treat this disease successfully.
In doing so, the researchers developed what they hope will be the first animal model for the disease that includes a severe bone depletion called humoral hypercalcemia of malignancy (HHM), a condition that can affect four out of every five ATLL patients and shorten their lives.
The study is published in the online edition of the journal Cancer Research.
“These ATLL tumors secrete proteins that also cause the bones in these patients to weaken and resorb,” explained Thomas Rosol, professor of veterinary biosciences and dean of the College of Veterinary Medicine at Ohio State University.
“When that happens, the amount of calcium in the blood can build up to toxic levels.” So killing the cancer cells in these patients is only half the battle, he says. “We have to stop the resorption of bone and the release of calcium that the cancer causes.”
Earlier tissue culture studies on a new anticancer drug, PS-341, showed promise in attacking the cancer cells but before now, an effective animal model wasn't available for researchers to use that included HHM's calcium buildup.
Rosol and his team turned to a combination of PS-341 and zoledronic acid, a form of bisphosphonate that is widely used now to combat the bone loss of osteoporosis and other diseases.
They would then test the two drugs, separately and combined, in a group of specialized mice that had been injected with ATLL tumor cells.
“We can inject these tumor cells into the abdomen of the mice and they will grow in the animals' lymph nodes,” explained Rosol, “but normally, you can't detect the extent of the animal's disease until the cancer is in its later stages.”
To solve this, Rosol's team took a novel approach:
They took a gene responsible for a firefly's glow and genetically inserted it into these tumor cells. That gene produces the enzyme luciferase in the insects which, when combined with another compound, luciferin, causes the firefly's distinctive glow.
The mice then received these genetically modified tumor cells and the researchers injected luciferin into the animals. Cancer cells containing the luciferase would combine with the luciferin and glow in the dark, giving the team a clear picture of the extent of disease inside the animal.
“We put these mice inside a blackened chamber with a digital camera and then took their pictures. The only light present would be the light emitted by the cancer cells,” Rosol said.
“We just measured the light that we could see coming out of the animal – the more light, the more tumor growth; the less light, less tumor.”
He said that with the tumor cells emitting light, his team was able to gauge the volume of tumor cells in the animal's body. “It is amazingly sensitive and precise, letting us see to a level of only a few hundred cells,” he said. “That gives us a good method for monitoring the tumor cells.”
So when the researchers tested the effects of the two drugs, they found that the zoledronic acid halted the bone resorption, reducing the harmful calcium in the body, and that the anticancer drug PS-341 killed more than 95 percent of the ATLL cells.
“It was very effective against ATLL, eliminating almost all of the tumor cells,” Rosol said.
What they didn't expect was that in some mice treated only with the zoledronic acid and not with the PS-341 anticancer drug, the zoledronic acid reduced some of the cancer growth as well.
“We have no idea why some animals responded in this way while others didn't,” Rosol said, “but the next step may be to try to understand the processes involved in how the tumor cells cause the resorption of the bones and the release of calcium.
“Hopefully, we'll soon see this work tested in human clinical trials and that perhaps will lead to a treatment for this disease.”
Source: Ohio State University