Researchers discover a method for clamping down on a cancer-promoting enzyme
There are many pathways that allow an errant gene to turn a cell cancerous, and a number of these pathways go through a single enzyme called the p21-activated kinase 1, or PAK1.
Researchers at Fox Chase Cancer Center have now identified a molecule capable of shutting down PAK1 before the enzyme becomes active. Previous studies have linked PAK1 activity with breast cancer and have shown the enzyme is important in pathways involving the ras oncogene, which is thought to cause up to 30 percent of all cancers.
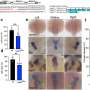
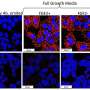
In the April 24 issue of the journal Chemistry & Biology, the researchers detail how the molecule, called IPA-3, was found from a screen of nearly 33,000 small molecules, and could serve as a basis for future breast cancer or cancer therapeutics. Cell-based studies using IPA-3 confirm that the molecule is capable of blocking signaling by the PAK1 pathway.
“Previous work suggested that hyperactive signaling by PAK1 can contribute to the growth of tumors, but the trick is how to selectively block PAK1 without damaging similar enzymes that are crucial for healthy cellular function,” said lead investigator Jeffrey R. Peterson, Ph.D, an associate member of Fox Chase. “IPA-3 represents a proof-of-principle, illustrating a new and highly selective approach to targeting PAK1.”
PAK1, like all kinases, is an enzyme that regulates other proteins by attaching an energetic molecule to them in a process known as phosphorylation. The “active site” where the phosphorylation reaction occurs is an attractive target for drug development, since blocking the active site would deactivate the enzyme. Unfortunately, the active site of PAK1 shares a molecular architecture similar to that found in many other kinase enzymes. Previous attempts to inhibit the PAK1 active site chemically have also resulted in inhibiting PAK1-related enzymes, with toxic consequences.
Instead of finding another molecule that binds to the active site, Peterson and his Fox Chase colleagues looked for new molecules that inactivate PAK1 in other ways. The cancer drug Gleevec, for example, is unusually selective for its target by binding to a region outside of the active site that is less common among kinases.
“Many other kinases, including PAK1, have unique regions outside the active site that mediate important facets of their function such as localization, substrate recruitment, or regulation,” Peterson says. “We wondered whether these regions might offer other places for molecules to bind and inhibit PAK1 without affecting other enzymes.”
According to Peterson, IPA-3 achieves high selectivity for PAK1 by taking advantage of a unique self-regulating region of the enzyme. The PAK1 protein has an auto-regulatory arm, a structure that PAK1 folds over its own active site when the enzyme is not in use. Their findings suggest that IPA-3 binds to the protein when it is in the closed configuration, which then prevents PAK1 from becoming active.
“It is like when the Steve Irwin would subdue a crocodile, he would tape its jaws closed to keep it from biting,” Peterson says. “Likewise, IPA-3 latches onto PAK1 in a way that prevents PAK1 from exposing its active site.”
Peterson and his colleagues, found IPA-3 by screening a library of over 33,000 small molecules for their ability to block phosphorylation by pure PAK1 protein. Any small molecules that blocked PAK1 were noted and were then ranked by potency, reproducibility and commercial availability. IPA-3 came out ahead of the others through this winnowing process, and the researchers then tested IPA-3 to demonstrate that it could also inhibit PAK1 activity inside living cells.
The Fox Chase researchers believe that IPA-3 represents a promising new strategy for creating therapeutics that inhibit PAK1 by mimicking the way cellular enzymes self-regulate in real life, but the IPA-3 molecule itself is not suitable as a therapeutic in its current form. “IPA-3 requires further experimental study and refinement before it could become a working drug for humans,” Peterson says.
Source: Fox Chase Cancer Center