Toward an effective treatment for a major hereditary disease
Scientists are reporting a key advance toward developing the first effective drug treatment for spinal muscular atrophy (SMA), a genetic disease that involves motor neuron loss and occurs in 1 out of every 6,000 births. SMA is the leading cause of hereditary infant death in the United States. The study is scheduled for publication online Oct. 8 by ACS Chemical Biology.
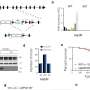
Mark E. Gurney, Jill Jarecki, and colleagues note that SMA is caused by a defective gene, SMN1, which fails to produce sufficient amounts of a key protein, called SMN (survival motor neuron), needed for normal motor neuron development. Scientists have screened more than 550,000 compounds in the search for a new SMA drug.
Recent research pointed to a group of compounds called C5-quinazolines that can boost SMN2 activity, a uniquely existing back-up gene for SMN1. In doing so, they showed promise for treating SMA by producing increased amounts of the needed protein.
In the new study, researchers identified exactly how these promising compounds work, a key step in moving forward toward medical use. They found that the substance targets a normal cellular protein, DcpS, involved in mRNA metabolism whose inhibition causes increased SMN expression. The finding could help guide the development of the first effective drugs for treating SMA and also lead to second generation drugs targeting this enzyme, the researchers say.
"The results outlined in the paper and carried out in collaboration with Families of SMA, deCODE chemistry & biostructures, Invitrogen Corporation, and Rutgers University represent a new understanding of the physiological mechanisms that can increase SMN expression and will allow us to move forward in advancing potential treatments for it, says Jill Jarecki, Ph.D., Research Director at Families of SMA.
Source: ACS