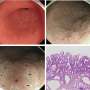
Researchers describe how chronic inflammation can lead to stomach cancer
A multi-center research team, led by Columbia University Medical Center, has uncovered a major contributor to the cause of stomach cancer – the second leading cause of cancer-related mortality in the world. The team described for the first time, that elevated levels of a single proinflammatory cytokine, an immune system protein called interleukin-1 beta (IL-1β), can start the progression towards stomach cancer. These results are published in the Nov. 4, 2008 issue of Cancer Cell. The researchers hope to use this finding to develop ways to block this process, thereby preventing cancer from developing.
"This study shows that accumulation of IL-1β, which is induced by infection with the bacterium Helicobacter pylori (H. pylori) in the gastrointestinal tract, is a significant contributor to the onset of stomach cancer," said lead author Timothy C. Wang, M.D., chief of the Division of Digestive and Liver Diseases and the Dorothy L. and Daniel H. Silberberg Professor of Medicine at Columbia University College of Physicians and Surgeons. "We show in this study that IL-1β works by activating a type of white blood cell known as myeloid derived suppressor cells (MDSCs), which in our study appeared to be strongly pro-inflammatory. Blocking IL-1β or the myeloid (MDSCs) cells may represent a potential strategy to prevent stomach cancer."
Previous research has shown that stomach cancer is strongly linked to chronic inflammation, and that infection with H. pylori may trigger the chronic inflammation that can lead to malignancy, but it was not known exactly how. While H. pylori infection is extremely prevalent, only a small minority (less than one percent) of infected individuals will, after many years, go onto develop stomach cancer. Previous research had linked H. pylori infection to the overexpression of IL-1β, and the susceptibility to gastric cancer to high-expressing IL-1 genotypes [Nature 2000;404:398-404], so Dr. Wang and his research team developed a transgenic mouse model in order to investigate the specific role of IL-1β in gastric carcinogenesis.
Results demonstrated that the overexpression of IL-1β in the stomach mobilizes the recruitment of MDSCs – initiating the progression of gastric inflammation into cancer. Furthermore, these findings help to explain why only a small percentage of those with H. pylori infection go onto develop stomach cancer – a genetic predisposition for high expression levels of proinflammatory cytokines.
Stomach Cancer is One of the Most Deadly & Common Cancers Worldwide
Stomach (gastric) cancer is the second (after lung cancer) most common cause of cancer-related mortality worldwide with 900,000 deaths this year. Stomach cancer is much more common in South America, Japan, Korea and Iceland than in the United States, which represents just two percent (25,500) cases of all new stomach cancer diagnosed yearly. It is associated with a diet that is high in salt and low in fruits and vegetables, as well as with smoking, and is more common in men. Infection with the bacterium Helicobacter pylori (H. pylori) is the main risk factor in about 80 percent or more of stomach cancers.
H. pylori is typically acquired in childhood through person to person transmission, and the bacterium lives within the stomach just above the stomach cells, where it induces a mild inflammatory response known as gastritis. H. pylori infection is generally associated with low socioeconomic status and poor hygiene. New H. pylori infection is gradually disappearing from most industrialized countries such as the United States and is now seen predominantly in underdeveloped countries, particularly in Asia and South America. H. pylori infection can lead to both stomach cancer and stomach ulcers but in the vast majority (more than 80 percent) of infected people, it causes no health problems.
Source: Columbia University