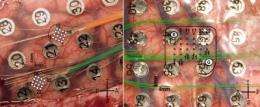
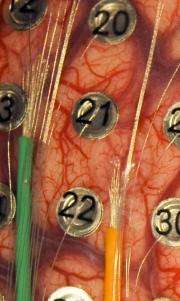
A new University of Utah study shows that arrays of tiny electrodes placed between the skull and the brain can accurately detect brain signals that command arm movements -- raising hope for longer-lived devices to help amputees control bionic arms and paralyzed people operate computers with their thoughts. These two images show two kinds of microelectrode arrays, known and microECoGs, that were placed on the brains of severe epilepsy patients. The patients already had parts of their skulls removed temporarily for placement of larger ECoG electrodes, which are used to locate and treat the brain area responsible for their seizures. These larger, metallic, button-like electrodes are numbered in both images. The left image also shows two microECoG arrays, each with 16 microelectrodes connected to microwires that pass through the orange and green tubes. Because the arrays are made of fine wires embedded in clear silicon, photo-editing software was used to draw in their outlines in both images. The right image shows one microECoG array with 32 individual microelectrodes, connected with microwires entering via a clear tube from the bottom of the image. The green wires are connected to the large, conventional ECoG electrodes. Credit: University of Utah Department of Neurosurgery
Experimental devices that read brain signals have helped paralyzed people use computers and may let amputees control bionic limbs. But existing devices use tiny electrodes that poke into the brain. Now, a University of Utah study shows that brain signals controlling arm movements can be detected accurately using new microelectrodes that sit on the brain but don't penetrate it.
"The unique thing about this technology is that it provides lots of information out of the brain without having to put the electrodes into the brain," says Bradley Greger, an assistant professor of bioengineering and coauthor of the study. "That lets neurosurgeons put this device under the skull but over brain areas where it would be risky to place penetrating electrodes: areas that control speech, memory and other cognitive functions."
For example, the new array of microelectrodes someday might be placed over the brain's speech center in patients who cannot communicate because they are paralyzed by spinal injury, stroke, Lou Gehrig's disease or other disorders, he adds. The electrodes would send speech signals to a computer that would covert the thoughts to audible words.
For people who have lost a limb or are paralyzed, "this device should allow a high level of control over a prosthetic limb or computer interface," Greger says. "It will enable amputees or people with severe paralysis to interact with their environment using a prosthetic arm or a computer interface that decodes signals from the brain."
The study is scheduled for online publication July 1 in the journal Neurosurgical Focus.
The findings represent "a modest step" toward use of the new microelectrodes in systems that convert the thoughts of amputees and paralyzed people into signals that control lifelike prosthetic limbs, computers or other devices to assist people with disabilities, says University of Utah neurosurgeon Paul A. House, the study's lead author.
"The most optimistic case would be a few years before you would have a dedicated system," he says, noting more work is needed to refine computer software that interprets brain signals so they can be converted into actions, like moving an arm.
An Advance over the Penetrating Utah Electrode Array
Such technology already has been developed in experimental form using small arrays of penetrating electrodes that stick into the brain. The University of Utah pioneered development of the 100-electrode Utah Electrode Array used to read signals from the brain cells of paralyzed people. In experiments in Massachusetts, researchers used the small, brain-penetrating electrode array to help paralyzed people move a computer cursor, operate a robotic arm and communicate.
Microwires emerging from the green and orange tubes connect to two arrays of 16 microelectrodes. Each array is embedded in a small mat of clear, rubbery silicone. The mats are barely visible in this image. These microelectrode arrays sit on the brain without penetrating it, a step toward longer-lived, less invasive versions of "neural interfaces" that in recent experiments elsewhere have allowed paralyzed people to control a computer cursor with their thoughts. The new microelectrode arrays were placed in two patients at the University of Utah who already were undergoing brain surgery for severe epilepsy. The larger, numbered, metallic electrodes are used to locate the source of epileptic seizures in the brain, so the patients allowed the microelectrodes to be placed on their brains at the same time. Credit: University of Utah Department of Neurosurgery
Meanwhile, researchers at the University of Utah and elsewhere are working on a $55 million Pentagon project to develop a lifelike bionic arm that war veterans and other amputees would control with their thoughts, just like a real arm. Scientists are debating whether the prosthetic devices should be controlled from nerve signals collected by electrodes in or on the brain, or by electrodes planted in the residual limb.
The new study was funded partly by the Defense Advanced Research Projects Agency's bionic arm project, and by the National Science Foundation and Blackrock Microsystems, which provided the system to record brain waves.
House and Greger conducted the research with Spencer Kellis, a doctoral student in electrical and computer engineering; Kyle Thomson, a doctoral student in bioengineering; and Richard Brown, professor of electrical and computer engineering and dean of the university's College of Engineering.
Microelectrodes on the Brain May Last Longer than Those Poking Inside
Not only are the existing, penetrating electrode arrays undesirable for use over critical brain areas that control speech and memory, but the electrodes likely wear out faster if they are penetrating brain tissue rather than sitting atop it, Greger and House say. Nonpenetrating electrodes may allow a longer life for devices that will help disabled people use their own thoughts to control computers, robotic limbs or other machines.
"If you're going to have your skull opened up, would you like something put in that is going to last three years or 10 years?" Greger asks.
"No one has proven that this technology will last longer," House says. "But we are very optimistic that by being less invasive, it certainly should last longer and provide a more durable interface with the brain."
The new kind of array is called a microECoG - because it involves tiny or
"micro" versions of the much larger electrodes used for electrocorticography, or ECoG, developed a half century ago.
For patients with severe epileptic seizures that are not controlled by medication, surgeons remove part of the skull or cranium and place a silicone mat containing ECoG electrodes over the brain for days to weeks while the cranium is held in place but not reattached. The large electrodes - each several millimeters in diameter - do not penetrate the brain but detect abnormal electrical activity and allow surgeons to locate and remove a small portion of the brain causing the seizures.
ECoG and microECoG represent an intermediate step between electrodes the poke into the brain and EEG (electroencephalography), in which electrodes are placed on the scalp. Because of distortion as brain signals pass through the skull and as patients move, EEG isn't considered adequate for helping disabled people control devices.
University of Utah neurosurgeon Paul A. House, shown here in an operating room, led a study showing that brain signals controlling arm movements can be detected accurately using new microelectrodes that sit on the brain but don't penetrate it. The tiny electrodes should last longer than those that poke into brain tissue, and may extend the useful lives of experimental devices to help amputees and paralyzed people operate robotic limbs or use thoughts to control a computer. Credit: Kelly Johnson, University of Utah
The regular-size ECoG electrodes are too large to detect many of the discrete nerve impulses controlling the arms or other body movements. So the researchers designed and tested microECoGs in two severe epilepsy patients who already were undergoing craniotomies.
The epilepsy patients were having conventional ECoG electrodes placed on their brains anyway, so they allowed House to place the microECoG electrode arrays at the same time because "they were brave enough and kind enough to help us develop the technology for people who are paralyzed or have amputations," Greger says.
The researchers tested how well the microelectrodes could detect nerve signals from the brain that control arm movements. The two epilepsy patients sat up in their hospital beds and used one arm to move a wireless computer "mouse" over a high-quality electronic draftsman's tablet in front of them. The patients were told to reach their arm to one of two targets: one was forward to the left and the other was forward to the right.
The patients' arm movements were recorded on the tablet and fed into a computer, which also analyzed the signals coming from the microelectrodes placed on the area each patient's brain controlling arm and hand movement.
The study showed that the microECoG electrodes could be used to distinguish brain signals ordering the arm to reach to the right or left, based on differences such as the power or amplitude of the brain waves.
The microelectrodes were formed in grid-like arrays embedded in rubbery clear silicone. The arrays were over parts of the brain controlling one arm and hand.
The first patient received two identical arrays, each with 16 microelectrodes arranged in a four-by-four square. Individual electrodes were spaced 1 millimeter apart (about one-25th of an inch). Patient 1 had the ECoG and microECoG implants for a few weeks. The findings indicated the electrodes were so close that neighboring microelectrodes picked up the same signals.
So, months later, the second patient received one array containing about 30 electrodes, each 2 millimeters apart. This patient wore the electrode for several days.
"We were trying to understand how to get the most information out of the brain," says Greger. The study indicates optimal spacing is 2 to 3 millimeters between electrodes, he adds.
Once the researchers develop more refined software to decode brain signals detected by microECoG in real-time, it will be tested by asking severe epilepsy patients to control a "virtual reality arm" in a computer using their thoughts.
Source: University of Utah (news : web)