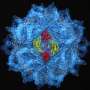
Structural biologists at the University of Pittsburgh School of Medicine have described the architecture of the complex of protein units that make up the coat surrounding the HIV genome and identified in it a "seam" of functional importance that previously went unrecognized. Those findings, reported today in Cell, could point the way to new treatments for blocking HIV infection.
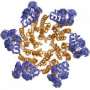
The researchers used a combination of nuclear magnetic resonance and cryoEM, which are standard structural biology tools, to see both the overall shape and the atomic details of capsid protein (CA) assembly. It takes about 1,500 copies of CA to make the coat, or capsid, that surrounds the genome of the AIDS virus.
"This strategy allowed us to see both the forest and the trees," explained study co-author Peijung Zhang, Ph.D., assistant professor in the Department of Structural Biology, Pitt School of Medicine. "Knowing what the CA protein looks like and how the capsid is built will allow scientists to rationally design therapeutic compounds that interfere with assembly of the protein and affect its function."
Capsid proteins, and particularly the interfaces or seams where one connects to another, are very important for assembling and disassembling the HIV coat, said senior author Angela Gronenborn, Ph.D., chair, Department of Structural Biology, and director, University of Pittsburgh Center for HIV Protein Interactions. The study indicates that these seams provide the flexibility to dismantle the coat efficiently after viral entry into the host and to put it back together when new viruses emerge from the cell.
"Our lab experiments show that if we replace a few of the pivotal stitches in the seam by mutation, the resulting viruses are less infectious or even non-infectious," Dr. Gronenborn said. "The capsid, and therefore the virus, can no longer function properly."
Source: University of Pittsburgh Schools of the Health Sciences (news : web)