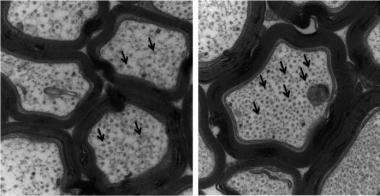
Microtubule (arrows) density in optic nerve of tau mice with EpoD treatment (top) is significantly higher compared to optic nerve in untreated (bottom) mice. Credit: Bin Zhang, MD, PhD, University of Pennsylvania School of Medicine.
(PhysOrg.com) -- Finding a drug that can cross the blood-brain barrier is the bane of drug development for Alzheimer’s disease and other neurological disorders of the brain. A new Penn study, published this week in the Journal of Neuroscience, has found and tested in an animal model of Alzheimer's disease a class of drug that is able to enter the brain, where it stabilizes degenerating neurons and improves memory and learning.
In the normal brain, the protein tau plays an important role in stabilizing structures called microtubules in nerve cells, which serve as tracks upon which cellular material is transported. In Alzheimer’s disease and related disorders, tau becomes insoluble and forms clumps in the brain. One consequence of these aggregates is a depletion of normal tau, resulting in destabilization of the microtubule tracks that are critical for proper nerve-cell function.
Senior authors Virginia M.-Y. Lee, PhD, director of the Center for Neurodegenerative Disease Research (CNDR), and John Trojanowski, MD, PhD, director of the Institute on Aging and CNDR co-director, introduced the concept of using microtubule-stabilizing drugs over 15 years ago to counteract tangles of tau and compensate for the loss of normal tau function. Kurt Brunden, PhD, director of Drug Discovery at CNDR and Bin Zhang, MD, PhD, senior research investigator, are the first authors on this study from the University of Pennsylvania School of Medicineand the School of Arts and Sciences.
In 2005, the CNDR researchers showed that the anti-cancer drug paclitaxel (Taxol) could improve spinal cord nerve function in mice with tau tangles in their brains, after the drug was absorbed at nerve termini in muscle. “However, paclitaxel and related drugs do not cross the blood-brain barrier” notes Brunden. “So we surveyed a number of additional microtubule-stabilizing agents and discovered that the epothilone class, and in particular epothilone D, readily entered and persisted in the brain.”
“The positive effect of epothilone D on the function of axons and on cognition, without the onset of side-effects offers hope that this class of microtubule-stabilizing drugs could progress to testing in Alzheimer patients in the near future,” says Lee.
“There are very few tau-focused drugs in clinical trials now for Alzheimer’s disease,” says Trojanowski. “While we and others have urged that pharmaceutical companies should not put all of their eggs in one drug basket to ensure the highest likelihood of finding disease-modifying therapies for Alzheimer’s, we hope this successful mouse study of a tau drug will encourage more pharmaceutical companies to pursue programs on tau-focused drug discovery.”
Help from Sponges
The epothilones are microtubule-binding drugs derived from marine sponges and have been used as anti-cancer drugs because they prevent cells from dividing. They do this by keeping microtubules overly stabilized, which blocks cell division and causes cell death in rapidly dividing cells such as cancer cells. However, since nerve cells do not replicate or divide, they are immune to the toxic effects of microtubule-binding drugs.
In Alzheimer’s disease and other diseases with tau clumps in the brain, the hope is that a microtubule-stabilizing drug will restore the microtubule tracks to their original supportive structure. This led the researchers to give the tau mice epothilone D (epoD) to replace the now unavailable tau.
Indeed, epothilone D improved the brain function of tau mice, which have tau inclusions in their forebrain, degenerated axons, and broken microtubules. After treating three-month old male tau mice with a low dose of epoD once a week for three months, the mice showed increased numbers of microtubules and improved axon integrity, without notable side effects to organs and immune cells.
What’s more, epothilone D reduced deficits in memory and learning in the tau mice. “EpoD improves cognition in mice affected by neurodegenerative tau pathology. These findings suggest that epothilone D and other microtubule-stabilizing agents hold considerable promise as potential treatments for neurodegenerative diseases in humans,” says senior author Amos B. Smith III, PhD, the Rhodes Thompson Professor of Chemistry.
Provided by University of Pennsylvania School of Medicine