Genetic 'conductor' involved with new brain cell production in adults
A team of North Carolina State University researchers has discovered more about how a gene connected to the production of new brain cells in adults does its job. Their findings could pave the way to new therapies for brain injury or disease.
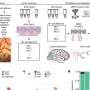
Most areas of the brain do not generate new brain cells, or neurons, after we are born. One exception is the olfactory bulb, the brain's scent processor, which continually produces new neurons. Dr. Troy Ghashghaei, assistant professor of neurobiology, had previously found a gene – known as Foxj1–connected to the production of an area inside the olfactory bulb where stem cells could form. Ghashghaei and his team discovered that Foxj1 was an "off switch" that told neuronal stem cells to stop reproducing and triggered the development of a stem cell "niche" in the olfactory bulbs.
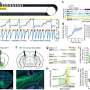
However, further experiments with newly developed genetically modified mice unexpectedly revealed that a fraction of Foxj1-expressing cells actually functioned as stem cells. But they only did so until the mouse reached the age equivalent of a human toddler, not throughout adulthood. In addition, the number of neurons generated by these cells was much lower than expected, which led to more questions about its function.
"Essentially, the experiments we did weren't giving us the numbers of neurons from Foxj1-expressing stem cells that we expected. We could have gotten disappointed with what may have been perceived as a road-block in our findings" says Ghashghaei. "If the gene was one that stem cells had to express in order to produce neurons, then we should have seen a greater number of neurons produced from the Foxj1-expressing stem cells. Instead, only about three percent of the olfactory neurons came from the Foxj1 stem cells. More importantly, we could not identify these unique neurons as belonging to known types of neurons in the olfactory system."
These findings and subsequent experimentation helped the team discover that in addition to being an off switch, the Foxj1 cellular lineage (i.e., Foxj1 expressing cells and their descendents) performs an important function as a "conductor," instructing the other stem cells in the olfactory bulb by secreting various molecules that affect the other stem cells' behavior and ensure their correct development into neurons. So a small number of Foxj1-expressing cells and their neuronal offspring direct other stem cells to continue reproducing, and may be telling them when to become functionally integrated neurons.
The research appears in the Journal of Neuroscience. Graduate students Benoit Jacquet, Huixuan Liang, and Guanxi Xiao, together with postdoctoral fellows Nagendran Muthusamy and Laura Sommerville contributed to the work.
"This finding is important because for the most part our brains cannot generate new neurons, nor can we efficiently use transplanted neurons to repair damage," Ghashghaei says. "Foxj1 expressing cells and their neurons seem to support zones within the brain where new neurons are created and integrated into existing neural circuits. If we can find out how to put these 'conductor' cells into other areas of the brain such as the spinal cord, it may lead to new cell-based therapies.
"This project took us on a roller-coaster ride – but the ending is a testament to the power of creative thinking and persistence in scientific inquiry – an achievement of which the clever and hardworking graduate students and postdoctoral fellows working on the problem should be very proud."
Ghashghaei's research is funded by the NIH and the American Federation for Aging Research. The Department of Molecular Biomedical Sciences is part of NC State's College of Veterinary Medicine.
More information: "Specification of a Foxj1-Dependent Lineage in the Forebrain Is Required for Embryonic-to-Postnatal Transition of Neurogenesis in the Olfactory Bulb" Authors: Benoit V. Jacquet, Nagendran Muthusamy, Laura J. Sommerville, Guanxi Xiao, Huixuan Liang, H. Troy Ghashghaei, North Carolina State University, Published: June 22, 2011 in Journal of Neuroscience.
Abstract
Establishment of a neural stem cell niche in the postnatal subependymal zone (SEZ) and the rostral migratory stream (RMS) is required for postnatal and adult neurogenesis in the olfactory bulbs (OB). We report the discovery of a cellular lineage in the SEZ-RMS-OB continuum, the specification of which is dependent on the expression of the forkhead transcription factor Foxj1 in mice. Spatially and temporally restricted Foxj1+ neuronal progenitors emerge during embryonic periods, surge during perinatal development, and are active only for the first few postnatal weeks. We show that the development of the unique Foxj1-derived lineage is dependent on Foxj1 expression and is required for overall postnatal neurogenesis in the OB. Strikingly, the production of neurons from Foxj1+ progenitors significantly declines after the early postnatal weeks, but Foxj1-derived neurons in the OB persist during adult periods. For the first time, our study identifies the time- and region-specific activity of a perinatal progenitor domain that is required for transition and progression of OB neurogenesis from the embryonic-to-postnatal periods.