Long-term correction of severe spinal muscular atrophy by antisense therapy
A new study from Cold Spring Harbor Laboratory (CSHL) reports surprising results that suggest that the devastating neuromuscular disease, spinal muscular atrophy (SMA), might not exclusively affect the motor neurons in the spinal cord as has long been thought. The new findings suggest that defects in peripheral tissues such as liver, muscle, heart, etc., might also contribute to the pathology of the disease in severely affected patients. The study, which also paves the way for a potential SMA drug to enter human trials by the end of the year, appears in Nature on October 6.
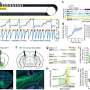
These insights stem from experiments that tested the new candidate drug, which the CSHL scientists helped develop, in a mouse model of very severe SMA. In this system, the candidate drug dramatically suppressed symptoms when simply injected under the animals' skin. "These systemic, or subcutaneous, injections, extended the lifespan of mice that have the equivalent of severe human SMA by 25-fold," reports CSHL's Professor Adrian Krainer, Ph.D., who led the CSHL team in collaboration with a group led by Dr. Frank Bennett of California-based Isis Pharmaceuticals.
"However, we have yet to determine whether these findings are unique to this animal model of severe SMA—and by extension, relevant only to the patients with the most severe disease —or if they will be valid in other SMA types that manifest with milder, less severe symptoms," cautions Dr. Bennett.
The No. 1 genetic cause of death among children under the age of two, SMA is caused by insufficient levels of the survival of motor neuron (SMN) protein due to mutations in the SMN1 gene. As a result, the spinal cord's motor neurons deteriorate and the muscles they control waste away. However, humans have a second gene, SMN2, which also produces SMN protein, but in small amounts due to a glitch that occurs during splicing – the molecular editing of RNA after it has been copied from DNA.
"Our strategy to decrease SMA severity was to boost SMN protein levels by fixing this splicing glitch," explains Krainer, a leader in the field of alternative splicing. When alternatively spliced, the same messenger RNA can yield different proteins, and mutations in splicing-regulatory sites can result in disease-causing protein deficiencies, as seen in SMA. Last year, his team used anti-sense oligonucleotides (ASOs)—small, chemically modified bits of RNA that can be custom-designed to redirect splicing—to improve SMA symptoms in mice with a mild form of the disease.
"Those experiments showed that ASOs injected directly into the fluid that surrounds the animals' brain and spinal cord could enhance SMN levels and improve symptoms," says Krainer. "They were based on the idea that SMN2 splicing had to be corrected in the motor neurons because these were the cells that were dying in SMA patients." But injecting ASOs into the brains of mice that have a severe form of SMA achieved a barely discernable increase in lifespan. This result prompted Krainer and his colleagues to wonder whether correcting SMN2 splicing in other tissues besides the brain and spinal cord could significantly improve the outcome.
"SMA has a very complex pathology that's still not fully understood," Krainer notes. "Patients with severe SMA have pathologies such as cardiovascular defects that extend beyond the central nervous system (CNS). So we began to explore whether boosting SMN levels outside the CNS could have a positive impact on muscle health and overall, and thereby alleviate the disease." Their hunch has been proved correct by the results described in the current paper.
Injecting ASOs systemically—i.e., under the skin—of mice with severe SMA a day after birth increased their life span from 10 days to an average of 108 days – a 10-fold increase. And increasing the dose of ASOs in systemic injections further resulted in a dramatic 25-fold increase in survival time. "These 'rescued' mice look and behave remarkably like normal control mice," says the study's lead investigator Yimin Hua, Ph.D.
Although the systemic ASO injections partially raised SMN levels in the CNS, "most of the ASOs injected via the skin were internalized in the liver, which is where the most robust correction in SMN2 splicing and the boost in SMN protein production occurred," reports Krainer. This observation, as well as the relatively smaller sizes of mice with severe SMA—which indicated growth problems—prompted the scientists to explore a liver-regulated growth hormone pathway involving a protein called IGF-1 (Insulin-like Growth Factor).
Greatly reduced or undetectable in untreated mice with severe SMA, the levels of IGF-1 were restored to normal following ASO treatment. "We don't yet know if this will be clinically relevant in humans because we are yet to find out whether children with severe SMA also have low IGF-1 levels," cautions Krainer. Clinical collaborations are now being planned to address this question.
The low IGF-1 levels seem to be due to insufficient levels of another protein, called IGF-acid labile subunit, whose role is to stabilize IGF-1 and extend its half-life in the bloodstream. The team has found that mice with severe SMA have reduced levels of this subunit's RNA and is now trying to determine why this occurs.
"While we know that SMA is caused by loss of function of the SMN1 gene, we don't know much about the downstream targets of SMN or how they contribute to the disease," said Michael Bender, Ph.D., who oversees Krainer's grant at the National Institute of General Medical Sciences of the National Institutes of Health. "By uncovering an effect on a key growth factor in the liver and underscoring the impact of rescuing the defect at the systemic level in a mouse model of SMA, this study brings us a step closer to a fuller understanding of SMA and offers a new approach to treating it."
More information: "Peripheral SMN restoration is essential for long-term rescue of a severe SMA mouse model," appears in Nature on October 6, 2011.