Study reveals potential of manganese in neutralizing deadly Shiga toxin
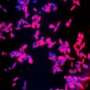
Carnegie Mellon University researchers have discovered that an element commonly found in nature might provide a way to neutralize the potentially lethal effects of a compound known as Shiga toxin. New results published in the Jan. 20 issue of Science by Carnegie Mellon biologists Adam Linstedt and Somshuvra Mukhopadhyay show that manganese completely protects against Shiga toxicosis in animal models.
Produced by certain bacteria, including Shigella and some strains of E. coli, Shiga toxin can cause symptoms ranging from mild intestinal disease to kidney failure. The findings could pave the way for future research aimed at creating an inexpensive treatment for infections caused by bacteria that produce the lethal Shiga toxin. Currently there is no treatment for such infections that afflict more than 150 million people each year, resulting in more than one million deaths worldwide.
Such infections are common in developing countries where it causes waterborne epidemics. It can be particularly deadly, especially in children, as it causes dysentery and severe hemorrhagic diarrhea, which cannot be adequately treated in areas without access to clean water. In developed countries, Shiga toxicosis is most common during foodborne outbreaks — like the widespread E. coli outbreak this past summer in Germany and Western Europe, where more than 3,700 people were infected and 45 died.
After entering the body, Shiga toxin is secreted by the infecting bacteria. It then attaches itself to a surface receptor on a cell's plasma membrane and enters the cell through a process called endocytosis. Normally, when a harmful substance enters a cell in this way, it's wrapped in a package called an endosome and sent directly to the cell's lysosome where it is degraded and discarded.
"That's exactly the process that Shiga toxin avoids. It would be neutralized if it were to get degraded, so it had to find some way to get out of being sent to the lysosome," said Linstedt, professor of biological sciences at Carnegie Mellon.
Linstedt and Mukhopadhyay discovered exactly how Shiga toxin avoided the lysosome as they were doing basic biological research to understand how components of the cell function. "If we weren't focused on answering fundamental biological questions, we wouldn't have made this discovery," Linstedt said.
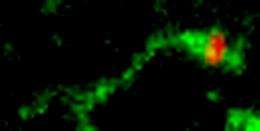
Fifteen years ago Linstedt discovered GPP130, a protein found in the Golgi apparatus, a kind of post office for the cell that sorts and packages molecules made in the endoplasmic reticulum and delivers them to their final destinations within the cell. GPP130, Linstedt found, didn't behave like most Golgi proteins. Rather than remaining in the Golgi, GPP130 constantly cycles to the endosomes and back to the Golgi. As it returns, it avoids the pathway that takes a substance to the lysosome to be degraded.
In Science, Linstedt and Mukhopadhyay report that Shiga toxin exploits this unique quality of GPP130 to its advantage. As it starts its return to the Golgi, GPP130 moves into a tubular extension off of the endosome's membrane and avoids being sent to the degradation pathway. Shiga toxin binds to GPP130, hitching a ride on a route that doesn't go to the lysosome. Instead, the toxin is carried to the Golgi apparatus and then to the endoplasmic reticulum, where it gets released into the cell's cytoplasm. It's here that it does its damage, shutting down protein synthesis and causing the cell to die.
"I knew that Shiga toxin was one of the key cargo molecules that bypass the lysosome as they go from the endosome to the Golgi apparatus, so I figured it would be a good marker to study in relation to GPP130. What I didn't realize was how profoundly dependent Shiga toxin was on GPP130," Linstedt said.
But the most serendipitous aspect of this discovery can be traced to a phone call made four years ago. Don Smith, a toxicologist at the University of California, Santa Cruz, who was studying manganese toxicity, noticed that GPP130 was sensitive to manganese. He called Linstedt who began to do a series of experiments on how manganese affects GPP130. Around this time Mukhopadhyay joined Linstedt's lab for his post-doctoral training. He quickly discovered that as concentrations of manganese rise inside the cell, GPP130 changes its pathway and goes directly to the lysosome where it is degraded.
Because Shiga toxin was dependent on GPP130 and manganese caused loss of GPP130, Linstedt and Mukhopadhyay decided to see whether manganese would protect against Shiga toxin infection. In cell cultures, manganese treatment yielded an almost 4,000-fold increase in the amount of Shiga toxin required to induce cell death. In a mouse model, mice exposed to a high dose of Shiga toxin and treated with non-toxic doses of manganese were 100 percent resistant to the toxin.
By introducing manganese, Linstedt and Mukhopadhyay were able to remove Shiga toxin's vehicle for avoiding degradation — GPP130. The researchers feel that this could be a promising treatment for neutralizing the effects of Shiga toxin in humans.
"Manganese is inexpensive. While Shiga toxin infection affects people in the developed world, it affects far more people in the developing world. An inexpensive, accessible treatment — not a designer drug — is the ideal solution," Linstedt said. "We know the toxicity levels of manganese in humans; we know ways to administer it. While further testing is needed to determine if manganese is a suitable treatment for humans, I'm optimistic that trials should move forward quickly."
The researchers also believe that manganese might be able to be used in conjunction with antibiotics. Currently, if an infected person is given an antibiotic, the antibiotic kills the bacteria (E. coli or Shigella) that produce the toxin. This releases the toxin in larger amounts and causes a higher percentage of patients to die. Linstedt believes that they can use manganese to block the toxin and an antibiotic to kill the bacteria, making for an extremely effective therapy.
The Carnegie Mellon researchers plan to continue their research by using situations that more closely mimic Shiga toxin infection in humans, and by testing their antibiotic/manganese combination therapy in mouse models. The researchers also will pay attention to manganese toxicity. High doses of manganese can have severe neurotoxic effects. The amounts used in the current study were low enough that they didn't cause any side effects in the mouse models.