The influence of the mother: Maternal epigenetic inheritance
(Medical Xpress) -- A study published in Genes and Development from scientists at the Friedrich Miescher Institute for Biomedical Research pinpoints the importance of maternal epigenetic influences during early embryogenesis in mammals. A chromatin regulatory complex in the oocyte ensures that the proper luggage of maternal transcripts and chromatin structures control the first steps in the formation of an embryo. In the absence of this epigenetic regulator the embryo fails to develop correctly.
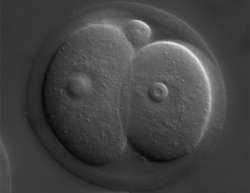
At the beginning of life, two highly specialized cells, the sperm and the egg, fuse to form a single totipotent cell that is capable of forming all the cell types in the body. How the cells achieve this complete switch from extreme specialization to potent all-rounder is to date not clear. However, an increasing amount of evidence points to an important role for heritable information that is not encoded by the DNA sequence, but is carried as epigenetic modification on the basic blueprint called the genome. Scientists have shown that the acquisition of totipotency, a capacity to form all the cells in the body, is accompanied by chromatin remodeling in the parental genomes, changes in the maternal transcriptome and proteome, and the novel activation of the newly formed zygotic genome. How these processes are governed, whether there is a differential inheritance from the maternal and paternal genomes in the form of epigenetic marks, and how many changes arise from reprogramming in the fertilized egg itself, are still unclear.
Antoine Peters and his team at the Friedrich Miescher Institute for Biomedical Research are addressing the role of maternal and paternal inheritance in the changes that occur during the conversion to totipotency. In a recent publication in Genes and Development they showed that two chromatin regulatory proteins, which form a key part of the Polycomb Group complex called PRC1, specify the maternal contribution to the earliest stages in the development.
During the development of an oocyte, the PRC1 complex ensures that appropriate genes are silenced or actively transcribed. The loss of two key components of PRC1, Ring1 and Rnf2, in oocytes led to massive transcriptional mis-regulation during oocyte growth and, after fertilization, provoked a developmental arrest at the two-cell stage of embryogenesis. This argues that the integrity of the PRC1 complex, and probably the ubiquitin conjugation that is mediated by Ring1 and Rnf2, ensures that the oocyte brings along the right maternal transcripts and chromatin structures to initiate the first steps in embryogenesis. Both cytoplasmic and chromosomal contributions from the maternal side, establish the developmental competence of early embryos.
“Part of the specialization taking place during the formation of the oocyte is devoted to ensuring the proper events in the earliest stages of embryogenesis,” comments Peters. “Our work suggests that inherited chromatin states and transcripts from the maternal side direct totipotency by regulating the transcriptional activation or silencing of the genome in the early embryo. This indicates that the newly formed embryo does not start from a clean slate. Epigenetic factors, which may be altered during the development of the oocytes, will have a strong influence on early steps in embryogenesis.”
More information: Posfai E, et al. (2012). Polycomb function during oogenesis is required for mouse embryonic development. Genes Dev. 2012 Apr 12. genesdev.cshlp.org/content/26/9/920.long