Protein pathways provide clues in leukemia research
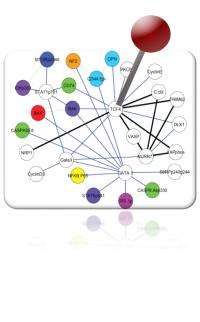
Scientists at Rice University and the University of Texas MD Anderson Cancer Center have successfully profiled protein pathways found to be distinctive to leukemia patients with particular variants of the disease.
Their research involved the creation of a new computational approach to identifying complex networks in protein signaling. They reported their work this month in the journal Proteomics.
With their new method, Rice bioengineer Amina Qutub, MD Anderson clinician and scientist Steven Kornblau and Rice graduate student Heather York analyzed more than a decade's worth of data. They identified patterns in the expression of 203 proteins studied in cells of acute myeloid leukemia (AML) patients and found nearly 700 protein pairs (from among thousands of possibilities) implicated in such networks.
The goal is to fine-tune therapy for patients, Qutub said. If it's possible to know the protein signaling pathways of particular types of leukemia, she said, then drugs may be found – or designed – to target the disease by blocking those pathways.
"Leukemia is not just one disease," said Qutub, who attended Rice as an undergraduate and is now an assistant professor of bioengineering based at Rice's BioScience Research Collaborative. "It's many, many diseases lumped into one category. You can't use a sledgehammer approach to treat it, though that's what clinicians have been doing."
"A Phillips head screwdriver does great if you've got a cross on your screw head," Kornblau said. "It doesn't work very well if you've got a slot. Companies have drugs that might work gangbusters in a small fraction of patients, but if you study a large aggregate, it doesn't look like a very active agent. Ideally, if it works in 5 percent of patients and you can pick them out ahead of time, you put them on the right drug and spare the other 95 percent from getting a drug that is not going to help them."
It's not unusual for leukemia patients to go into remission, Qutub said, but most suffer a relapse within two years. "We're trying to figure out what's so different about these patients, and whether we can enhance their therapy. The only way we know to do that is to figure out the protein signature."
Proteins are the workhorses of the cell. Created from blueprints stored in DNA, they pass chemical signals from one to another with instructions on just about every activity a cell is capable of. But when their basic coding is scrambled, they can just as easily cause a cell to self-destruct.
Qutub likened protein signaling pathways to city traffic. "You might take a train, drive a car, walk or take a bicycle, but eventually you would get to your location," she said. "In cancer patients, protein pathways seem to follow patterns. In normal cells, there are predominant pathways, and while you may have the same pathways in cancer cells, they might also have alternate routes – and take them, to varying degrees."
Specific routes proteins use to pass signals may be the key to determining their type, so it was worth investigating a treasure trove of data that had been gathered from MD Anderson patients, Qutub said.
The project began with a chance meeting by Qutub and Kornblau at a conference in Colorado on hypoxia. "Steve said, 'You're working with mathematical modeling of hypoxic response. I have a patient set I think you might be interested in.' That's how it started, and it took us down a very different road, using machine-learning algorithms and statistical techniques that hadn't been part of my core research but have now become integral to both this project and other research in the lab," Qutub said.
The World Health Organization had categorized AML patients by certain cell phenotypes, Qutub said, "but very little had been done at the protein level. Our clinical collaborators had started genetic and proteomic studies a while back and identified recurrent patterns of protein expression, some of which were associated with recurrent chromosomal changes like 'inv (16),' the inversion of chromosome 16, or 't(8;21),' which is the transpose of the 8 and 21 chromosomes." They are found in about 15 percent of patients with AML, she said.
"Patients with these two chromosomal changes respond more favorably to current AML therapy," Kornblau said, "and the genes from each of these genetic abnormalities are involved in the same transcription factor complex, creating a mechanistic link. But despite this mechanistic link, they differ in some clinical features and outcome."
Kornblau's data was precisely what Qutub and her group needed. They targeted inv (16) and t(8;21) to learn what protein pathways were common to both of these cytogenetic categories, what might explain their increased responsiveness and what changes were unique to each that might explain their clinical differences. From the mass of data – more than 20,000 possible protein pairs – they found that 678 pairs related to inv (16) and t(8;21) were significantly different compared with normal cells.
"There are some genetic markers that are definitely associated with a prognosis of leukemia," Qutub said. "And other things can cause leukemia -- acute myeloid in particular -- perhaps even chemotherapy itself. The way cancer patients are treated may be causing mutations and in turn triggering leukemic response."
With their analytical method tried and tested, Qutub and her team can now statistically group patients who present with particular protein pathways by age, gender, therapies and other criteria, and then take a closer look at protein signaling pathways in bone marrow-derived stem cells, a prime suspect in patients who relapse even after decades of remission. "We think something has to be unique about stem cells," she said.
The key problem with all cancers, including leukemia, is their plasticity, she said. "Cancer has multiple ways of getting to the same destination. You try one drug, and cancer's going to circumvent you and go a different way. What we're trying to say is, 'Here are all the possible ways,' so we can begin to target the intersections."
More information: Read the abstract at onlinelibrary.wiley.com/doi/10 … c.201100491/abstract















