Researchers reveal different mechanisms of pain
Researchers at the University of Leeds have found a previously unknown mechanism through which pain is signalled by nerve cells – a discovery that could explain the current failings in the drug development process for painkillers and which may offer opportunities for a new approach.
The team, led by Dr Nikita Gamper of the University's Faculty of Biological Sciences, is investigating the difference between persistent pain, such as toothache, and pain that results from the increased sensitivity of nerves in injured or diseased tissue (for example when we touch inflamed skin), known as hyperalgesia.
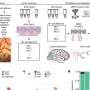
In research published online this week, (w/c 14 May) in Proceedings of the National Academy of Sciences (PNAS), Dr Gamper's team has discovered that these two types of pain are generated by the same nerves, but result from different underlying mechanisms.
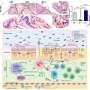
The project, funded jointly by the Wellcome Trust and the Medical Research Council, investigated the painful effects of two substances that cause local inflammation: bradykinin and substance P. Both substances bind to specific receptors on nerve cells, generating signals to the central nervous system. Because the receptors are from the same family, it has always been presumed they stimulate the same signalling pathway.
However, the team found that each receptor produces different signals; the one associated with bradykinin causing both hyperalgesia and persistent pain, whereas the one associated with substance P only caused hyperalgesia.
"Dr Gamper says: "Pain originates from a series of electrical signals sent by nerve cells in to the central nervous system and ultimately the brain. Despite much progress, we still don't know enough about the mechanisms by which these pain signals are generated. However, this research has shown that whilst the sensation of pain can be similar between various conditions, the underlying molecular mechanisms may in fact be very different."
Existing painkillers are 'non-specific', designed to generally dull the reception of these signals in the central nervous system, and some stronger pain killers can provoke unwanted side effects such as disorientation, drowsiness or nausea. So while the search for new better drugs is pressing, the lack of progress in developing targeted analgesics has led to several pharmaceutical companies dropping this area of research altogether.
"What's exciting about these findings is that substance P may actually suppress the activation of the pain sensing nerves themselves," says Dr Gamper.
"It's increasingly evident that current strategies for testing and validating new painkillers often do not take into account a possible difference in how pain signals are generated. For instance, drugs for persistent pain are often tested solely for their ability to reduce hyperalgesia, and as a result, some of the drugs that are effective in the lab, fail in subsequent clinical trials. These findings challenge current approaches in drug development research and may offer new strategies," he says.
More information: The paper, Reactive oxygen species are second messengers of neurokinin signaling in peripheral sensory neurons is published in the PNAS Online Early Edition in the week of May 14-18, 2012. doi:10.1073/pnas.1201544109