Parkinson's disease protein causes disease spread and neuron death in healthy animals
Understanding how any disease progresses is one of the first and most important steps towards finding treatments to stop it. This has been the case for such brain-degenerating conditions as Alzheimer's disease. Now, after several years of incremental study, researchers at the Perelman School of Medicine, University of Pennsylvania have been able to piece together important steps in how Parkinson's disease (PD) spreads from cell to cell and leads to nerve cell death.
Their line of research also informs the general concept that this type of disease progression is a common pathway for such other neurodegenerative diseases as Alzheimer's, Huntington's, progressive supranuclear palsy, and possibly amyotrophic lateral sclerosis (ALS).
The Penn team found that injecting synthetic, misfolded and fibrillar α-Synuclein (α-Syn) – the PD disease protein—into the brains of normal, "wild-type" mice recapitulates the cascade of cellular demise seen in human PD patients.
Parkinson's disease is characterized by abundant α-Syn clumps in neurons and the massive loss of midbrain dopamine-producing neurons. However, a cause-and-effect relationship between the formation of α-Syn clumps and neurodegeneration has been unclear.
In short, the Penn researchers found that, in healthy mice, a single injection of synthetic, misfolded α-Syn fibrils led to a cell-to-cell transmission of pathologic α-Syn proteins and the formation of Parkinson's α-Syn clumps known as Lewy bodies in interconnected regions of the brain. Their findings appear in this week's issue of Science. The team was led by senior author Virginia M.-Y Lee, PhD, director of the Center for Neurodegenerative Disease Research (CNDR) and professor of Pathology and Laboratory Medicine, and first author Kelvin C. Luk, PhD, research assistant professor in the CNDR.
The major significance of the paper is that it resolves the long-standing controversy about the role of α-Syn Lewy bodies in the degeneration of substantia nigra dopamine neurons, thereby sharpening the focus on Lewy bodies as targets for discovery of disease modifying therapy for Parkinson patients.
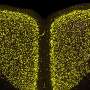
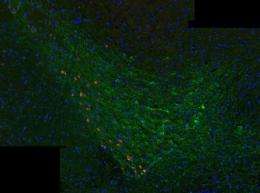
The α-Syn clumps caused progressive loss of dopamine neurons in the connected substantia nigra region of the brain. This finding was accompanied by reduced dopamine levels in the neurons of the striatum, which cause the movement disorder in Parkinson's patients.
The team saw α-Syn pathology in the wild-type mice one month after injection. After three months one sixth of dopamine-producing neurons were gone and after six months half of dopamine-producing neurons were gone. In addition, the injected wild-type mice did worse on motor skill tests of grip strength, balance, and co-ordination compared to controls. The experiment was ended before cognitive defects were detected, which is common in about 80 percent of Parkinson's patients during the course of the illness.
The recapitulation of the neurodegenerative demise of neurons establishes a mechanistic link between transmission of pathologic α-Syn and the cardinal features of Parkinson's disease – death of dopamine-producing neurons and the formation of α-Syn clumps.
Pathological Templates
Two years ago, the same Penn team found that small amounts of misfolded α-Syn can be taken up by healthy neurons, replicating within the nerve cells to cause neurodegeneration. The α-Syn protein is normally found in brain synapses that connect nerve cells and enable their communication. In time, α-Syn forms the characteristic Lewy bodies in the neurons of patients with PD and some other neurodegenerative disorders. They found that abnormal clumps of α-Syn formed by small fibrils act as "seeds" that induce normal α-Syn molecules to misfold and form aggregates. This scenario then propagates from neuron to neuron over time in the brain. The pathological α-Syn acts as a template to corrupt the normal α-Syn so it too becomes pathological and thereby spreads the disease from an affected neuron to a normal one, which then becomes diseased.
In earlier studies at other institutions, when fetal nerve cells were transplanted into the brains of PD patients, some of the transplanted cells developed Lewy bodies. This also suggested that the corrupted form of α-Syn could somehow be transmitted from diseased neurons to healthy ones. In a study published earlier in 2012 in the Journal of Experimental Medicine, the Penn team showed that extracts of brain tissue from a PD mouse model, as well as synthetically produced α-Syn fibrils, injected into young, symptom-free transgenic mice that were engineered to overexpress α-Syn led to spreading of α-Syn pathology. By three months after a single injection, neurons containing abnormal α-Syn clumps were detected throughout the mouse brains. The inoculated mice died between 100 to 125 days post-inoculation, short of their typical two-year life span.
In contrast, the current Science study was conducted using healthy, wild-type or non-transgenic mice. This means that since this PD was induced in a healthy mouse, the researchers' approach is now a much more compelling model for studying the most common form of PD - sporadic PD, which accounts for greater than 90 percent of patients with PD whose disease does not run in families.
In addition, by using isolated, synthetic misfolded α-Syn, which has same properties as natural α-Syn, and not brain tissue from PD patients or PD mice, the researchers were able to establish that it was the misfiolded α-Syn alone that unequivocally causes the pathology and progression of PD in the healthy mice. The team is now working on an antibody therapy in these mouse models to stop propagation of rogue misfolded α-Syn. What's more, both the cell culture and the mouse models will facilitate the identification of novel targets for PD therapy.
More information: "Pathological α-Synuclein Transmission Initiates Parkinson-like Neurodegeneration in Nontransgenic Mice," by K.C. Luk, Science, 2012.