Hypertension traced to source in brain
(Medical Xpress)—When the heart works too hard, the brain may be to blame, says new Cornell research that is changing how scientists look at high blood pressure (hypertension). The study, published in the Journal of Clinical Investigation in November, traces hypertension to a newfound cellular source in the brain and shows that treatments targeting this area can reverse the disease.
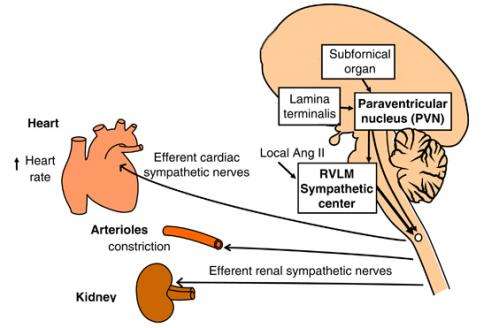
In what peer reviewers are calling "a new paradigm" for tackling the worldwide hypertension epidemic, this insight into its roots could give hope to the billion people it currently afflicts. Hypertension occurs when the force of blood against vessel walls grows strong enough to potentially cause such problems as heart attack, stroke and heart or kidney disease. The heart pumps harder, and often the hormone angiotensin-II (AngII) gets the pressure cooking by triggering nerve cells that constrict blood vessels.
"We knew the central nervous system orchestrates this process, and now we've found the conductor," said Robin Davisson, the Andrew Dickson White Professor of Molecular Physiology with a joint appointment at Cornell's College of Veterinary Medicine and Weill Cornell Medical College.
Two-thirds of Americans have hypertension, which is the leading cause of North America's No. 1 killer: heart disease, according to the Centers for Disease Control and Prevention.
Davisson's lab traced neurochemical signals back to endoplasmic reticulum (ER), the protein factory and stress-management control center in every cell. If something goes wrong in a cell, the ER activates processes to adapt to the stress. Long-term ER stress can cause chronic disease, and several stressors that ER responds to have been connected to hypertension. Davisson's lab found that high levels of AngII put stress on the ER, which responds by triggering the cascade of neural and hormonal signals that start hypertension.

But not just any cell's ER can conduct this complex orchestra. Those that can trigger the signal cascade are clustered near the bottom of the brain in a gatelike structure called the subfornical organ (SFO). Unlike most of the brain, the SFO hangs outside a protective barrier that keeps most circulating particles from entering the brain. The SFO can interact with particles like AngII that are too big to cross through and can also communicate with the brain's inner chambers.
This is good news for developing therapies—because the SFO sits outside the barrier, it can be reached through such normal treatment routes as pills or injections rather than riskier brain procedures. Davisson's lab showed that treatments that inhibit ER stress in the SFO can completely stop AngII-based hypertension and lower blood pressure to normal levels.
"Our work provides the first evidence that higher levels of AngII cause ER stress in the SFO, that this causes hypertension, and that we can do something about it," said Davisson. "This finding may also suggest a role for ER stress in hypertension types that don't involve AngII, like some spontaneous or genetic forms."
Inspired by the paradigm shift that this study has sparked, the editors of the Journal of Clinical Investigation published a commentary concluding that this discovery "opens new avenues for investigation and may lead to new therapeutic approaches for this disease."