Two studies reveal genetic variation driving human evolution
A pair of studies published by Cell Press on February 14th in the journal Cell sheds new light on genetic variation that may have played a key role in human evolution. The study researchers used an animal model to study a gene variant that could have helped humans adapt to humid climates, and they used whole-genome sequence data to identify hundreds of gene variants that potentially helped humans adapt to changing environmental conditions over time. The findings provide a road map for understanding human biological history as well as modern-day variability.
"There is an archaeological record hidden in our DNA that can help point us to the traits that have been critical in human survival, such as resistance to infectious diseases and new abilities to respond to different environments," says senior study author Pardis Sabeti of Harvard University and the Broad Institute. "The two studies have uncovered two intriguing human adaptive traits and demonstrate the ability to go from an unbiased genome scan to a novel hypothesis of human evolution."
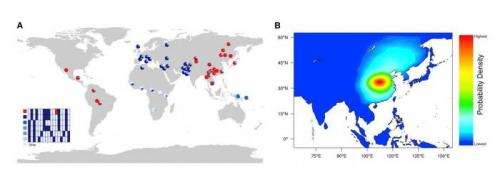
Sabeti and her team found that a previously reported variant of the EDAR gene, which arose in central China about 30,000 years ago, increased the number of sweat glands in genetically modified mice and had other effects not previously reported in humans; their discovery demonstrates that animal models can be used to study the biological changes expected to result from human genetic variation. This gene variant was also associated with an increase in the number of sweat glands in a present-day Han Chinese population. By enhancing sweating, this EDAR variant could have helped humans adapt to humid climates that may have existed in China 30,000 years ago.
In the accompanying study, the researchers used data from the 1000 Genomes Project to analyze DNA sequence variations across the entire genome. They identified hundreds of gene variants that potentially contributed to human evolutionary adaptation. One of these variants, a mutation in the TLR5 gene, changed the immune responses of cells exposed to bacterial proteins, suggesting that this variant could confer a fitness advantage by protecting against bacterial infections. The comprehensive list of possible adaptive mutations driving recent human evolution provides the groundwork for future studies.
Together, the two studies represent a decisive shift for the field of evolutionary genomics, transitioning from hypothesis-driven to hypothesis-generating science. By moving from genome-wide scans to the characterization of adaptive mutations, it is possible to elucidate distinct mechanisms of evolution.
"These two studies are the product of work done in this area for over a decade but can only now be made possible with the major breakthroughs in genomic technology," Sabeti says. "I am struck by the ability of genomics to uncover the secrets of human history."
















