Embryonic stem cell transplant restores memory, learning in mice

For the first time, human embryonic stem cells have been transformed into nerve cells that helped mice regain the ability to learn and remember. A study at the University of Wisconsin-Madison is the first to show that human stem cells can successfully implant themselves in the brain and then heal neurological deficits, says senior author Su-Chun Zhang, a professor of neuroscience and neurology.
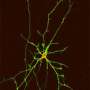
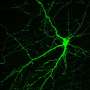
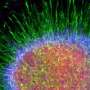
Once inside the mouse brain, the implanted stem cells formed two common, vital types of neurons, which communicate with the chemicals GABA or acetylcholine. "These two neuron types are involved in many kinds of human behavior, emotions, learning, memory, addiction and many other psychiatric issues," says Zhang.
The human embryonic stem cells were cultured in the lab, using chemicals that are known to promote development into nerve cells—a field that Zhang has helped pioneer for 15 years. The mice were a special strain that do not reject transplants from other species.
After the transplant, the mice scored significantly better on common tests of learning and memory in mice. For example, they were more adept in the water maze test, which challenged them to remember the location of a hidden platform in a pool.
The study began with deliberate damage to a part of the brain that is involved in learning and memory.
Three measures were critical to success, says Zhang: location, timing and purity. "Developing brain cells get their signals from the tissue that they reside in, and the location in the brain we chose directed these cells to form both GABA and cholinergic neurons."
The initial destruction was in an area called the medial septum, which connects to the hippocampus by GABA and cholinergic neurons. "This circuitry is fundamental to our ability to learn and remember," says Zhang.
The transplanted cells, however, were placed in the hippocampus—a vital memory center—at the other end of those memory circuits. After the transferred cells were implanted, in response to chemical directions from the brain, they started to specialize and connect to the appropriate cells in the hippocampus.
The process is akin to removing a section of telephone cable, Zhang says. If you can find the correct route, you could wire the replacement from either end.
For the study, published in the current issue of Nature Biotechnology, Zhang and first author Yan Liu, a postdoctoral associate at the Waisman Center on campus, chemically directed the human embryonic stem cells to begin differentiation into neural cells, and then injected those intermediate cells. Ushering the cells through partial specialization prevented the formation of unwanted cell types in the mice.
Ensuring that nearly all of the transplanted cells became neural cells was critical, Zhang says. "That means you are able to predict what the progeny will be, and for any future use in therapy, you reduce the chance of injecting stem cells that could form tumors. In many other transplant experiments, injecting early progenitor cells resulted in masses of cells—tumors. This didn't happen in our case because the transplanted cells are pure and committed to a particular fate so that they do not generate anything else. We need to be sure we do not inject the seeds of cancer."
Brain repair through cell replacement is a Holy Grail of stem cell transplant, and the two cell types are both critical to brain function, Zhang says. "Cholinergic neurons are involved in Alzheimer's and Down syndrome, but GABA neurons are involved in many additional disorders, including schizophrenia, epilepsy, depression and addiction."
Though tantalizing, stem-cell therapy is unlikely to be the immediate benefit. Zhang notes that "for many psychiatric disorders, you don't know which part of the brain has gone wrong." The new study, he says, is more likely to see immediate application in creating models for drug screening and discovery.
More information: Medial ganglionic eminence–like cells derived from human embryonic stem cells correct learning and memory deficits, DOI: 10.1038/nbt.2565