July 5, 2013 report
Research shows gut microbe populations stable over years, probably decades
(Medical Xpress)—The importance of the bacteria that live within our digestive tracts is just beginning to be fully realized, and while it has long been known that they assist in digestion and absorption of nutrients, little has been understood about the stability of the microbiota population. Now a U.S. study suggests the gut flora are relatively stable over several years and possibly over our entire lifetimes.
A team led by Jeffrey I. Gordon of the Center for Genome Sciences and Systems Biology at Washington University School of Medicine in St. Louis, developed a new method for investigating the composition of the gut microbiota over time. The method, called low-error amplicon sequencing (LEA-Seq) takes advantage of the fact that each strain of gut bacteria carries a slightly different variation in a gene, and these variations can therefore act as biological "barcodes" to identify the bacterial strains present in a sample. High error rates of other methods were avoided by making many more copies of the "barcoded" DNA fragments than usual so that they could be sequenced more times.
The study analyzed fecal microbiota obtained from 175 stool samples from 37 healthy women across the U.S. In 33 of the volunteers, samples were obtained 2-13 times with first and last samples taken up to 296 weeks apart. The other four volunteers were on a low-calorie liquid diet as part of an in-patient weight-loss study and were sampled around every 16 days for up to 32 weeks.
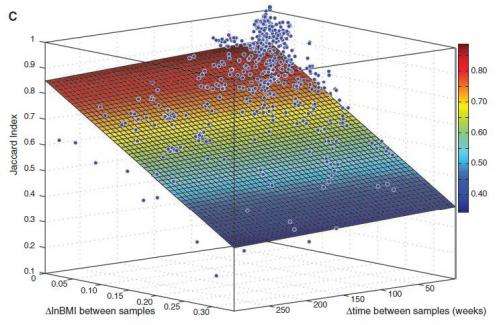
The results showed that in the 33 non-obese subjects the gut microbe population consisted of 195 ± 48 bacterial strains of 101 ± 27 species, and the composition remained stable over time, with over 70 percent of the same strains being found a year after the initial sampling, and over 60 percent after five years. The researchers also found that the most stable strains of bacteria were the most abundant.
The results followed a power law function, which when extrapolated, suggests the gut flora could remain fairly constant for decades, and possibly throughout our lives.
In the four patients being treated for obesity with a monotonous liquid diet, the microbiota were less stable, with some strains becoming much less common during the weight-loss period, and others becoming more abundant. These changes were greatest in those women who lost the most weight (up to 10 percent of their body weight).
Another aspect of the research was to look at the gut flora of related and unrelated individuals, and the results showed that the composition of the microbiota was more similar in relatives than in non-related people, even if the relatives did not live close to each other. These findings suggest that the bacterial populations were established early in life and then remained reasonably constant thereafter.
The results of the study show the bacteria in the human gut are remarkably constant and variations in their composition could affect our health and risk for disease throughout our lives. Co-author of the Science paper, Jeremiah J. Faith, suggested that in future a routine analysis of gut bacteria found in stool samples could be a regular part of a health checkup so that variations in the flora could be identified, as changes could be related to developing conditions, perhaps including autoimmune diseases.
More information: The Long-Term Stability of the Human Gut Microbiota, Science 5 July 2013: Vol. 341 no. 6141 DOI: 10.1126/science.1237439
Abstract
A low-error 16S ribosomal RNA amplicon sequencing method, in combination with whole-genome sequencing of >500 cultured isolates, was used to characterize bacterial strain composition in the fecal microbiota of 37 U.S. adults sampled for up to 5 years. Microbiota stability followed a power-law function, which when extrapolated suggests that most strains in an individual are residents for decades. Shared strains were recovered from family members but not from unrelated individuals. Sampling of individuals who consumed a monotonous liquid diet for up to 32 weeks indicated that changes in strain composition were better predicted by changes in weight than by differences in sampling interval. This combination of stability and responsiveness to physiologic change confirms the potential of the gut microbiota as a diagnostic tool and therapeutic target.
© 2013 Medical Xpress
















