July 30, 2013 feature
Patient, heal thyself: Functional blood vessels regenerated in vivo from human induced pluripotent stem cells
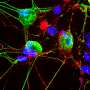
![In vivo imaging of hiPS cell-derived engineered blood vessels. (A) Multiphoton laser-scanning microscopy image of HS27-iPS-ECs (green) and 10T1/2 cells (blue) coembedded in a fibronectin/collagen I tissue-engineered vessel construct and inoculated in SCID mice in a cranial window. These cells developed functional perfused blood vessels [red, 1,1-dioctadecyl-3,3,3,3-tetramethylindodicarbocyanine perchlorate (DID)-labeled RBCs] in vivo (day 14). (B–D) Functional assessment of iPS cell-derived engineered blood vessels. (B) Perfused vessels engineered from HS27-iPS-ECs (green) imaged after injection of DiDRBCs (red) and Alexa 647-BSA (blue). (C) Map of RBC velocity quantified by recently established line and full-field RBC velocity-scanning techniques (45). Engineered vessels are well perfused. (D) Histogram of RBC velocity profile in iPS cell-derived vessels. HS27-iPS-ECs with 10T1/2 cells (blue bars), T1D-iPS-ECs with 10T1/2 cells (brown bars), and HS27-iPS-ECs with HS27-iPS-mesenchymal cells (green bars) are shown. (E) Map of vessel permeability to Alexa 647- labeled BSA quantified based on the extravasation of BSA from individual vessels over time. Engineered vessels have a segment with higher permeability compared with endogenous normal vessels. Multiphoton imaging was carried out on a custom-built multiphoton laser-scanning microscope using a confocal laser-scanning microscope body and a broadband femtosecond laser source. Imaging studies in A–C and E were performed at a magnification of 20×, using a 0.95-N.A. water immersion objective. Two-micron-thick optical sections were taken. The imaging field of view was 660 μm × 660 μm × 155 μm with a resolution of 1.3 μm × 1.3 μm × 2 μm. Copyright © PNAS, doi:10.1073/pnas.1310675110 Patient, heal thyself: Functional blood vessels regenerated in vivo from human induced pluripotent stem cells](https://scx1.b-cdn.net/csz/news/800a/2013/samuelfig2.jpg)
(Medical Xpress)—Vasculogenesis – the process of blood vessel formation through a de novo production of endothelial cells (ECs, or those forming a thin layer lining the interior surface of blood and lymphatic vessels) – is a vital tool in regenerative medicine, tissue engineering, and, in particular, the battle against vascular disease, the leading cause of mortality in the United States. (More than one in three Americans (36.9%) suffer from heart disease, and by 2030, an estimated 116 million people in the United States (40.5%) will have some form of cardiovascular disease.) More specifically, generating functional, long-lasting vasculogenic cells is a key but elusive component in human induced pluripotent stem (hiPS) research. Recently, however, researchers at Harvard University and Massachusetts General Hospital successfully generated endothelial cells from healthy donors' hiPS cells to form stable functional blood vessels in vivo. Moreover, they developed an approach to generate mesenchymal precursor cells (MPCs, or multipotent stromal, or connective tissue, cells that can differentiate into a variety of cell types including perivascular cells – another component of vessel wall) from hiPS cells in parallel, and also generated functional blood vessels in vivo using these endothelial and multipotent stromal derived cells from the same hiPS cell line. Beyond this, and in terms of clinical translation, the team successfully generated ECs and MPCs from Type 1 Diabetic patient-derived hiPS cell lines and also used them to generate blood vessels in vivo.
Drs. Rakesh K. Jain, Dai Fukumura and Rekha Samuel discussed the research they and their colleagues conducted, and the challenges they faced, with Medical Xpress. "In terms of the efficient generation of competent vasculogenic cells in hiPS cell-based regenerative medicine," Jain tells Medical Xpress, "we started from previously established methods for the generation of vasculogenic cells that were proven to work for other sources of stem/progenitor cells, such as human embryonic stem (hES) cells. However, from iPS cells, these methods failed to generate enough numbers of vasculogenic cells and/or to demonstrate in vivo functionality. Furthermore, we've demonstrated a significant variation between different iPS cell lines – and even within clones from the same person." In short, the researchers' novel techniques for deriving iPS cells towards vascular precursors highlight the importance of identifying vasculogenic clones that are capable of forming durable blood vessels in vivo.
Another challenge, Fukumura notes, was to monitor engineered blood vessel functionality during vessel development. "Most mouse models using iPS derived endothelial or mesenchymal cells to date relied on invasive approaches such as serial histological sections to examine blood vessels," he explains. "Such approaches don't permit real time vasculogenesis examination and require a large number of experimental animals to assess any kinetic changes." However, the researchers' proven track record of establishing and using state-of-the-art in vivo imaging techniques – a powerful combination of sophisticated so-called transparent window animal models, genetic and pharmacological labeling of target cell populations and molecules, advanced optical imaging devices (such as multiphoton laser-scanning microscopy used in this study), and computer-assisted image analysis algorithms – afforded them long-term, non-invasive, functional evaluation of iPS- derived blood vessels in living mice.
While the scientists had previously generated durable functional blood vessels in vivo using CD34 positive vasculogenic cells from hES cells1, this method turned out not to be optimal for iPS cells. Therefore, in the current study they decided to combine other vasculogenic markers – CD34 (a cell surface glycoprotein that mediated the attachment of stem cells directly to stromal cells), neuropilin 1 (NP1) and human kinase insert domain-containing receptor (KDR) – that have been successfully used to derive endothelial cells from hES cells. "To our knowledge," Samuel points out, "this is the first report demonstrating the use triple selection markers CD34, NP1 and KDR for the isolation of vasculogenic precursor cells from human induced pluripotent stem cells that are, most importantly, highly potent in an in vivo setting."
In the current study, the researchers also developed an efficient 2D culture system for hiPS cell-derived endothelial precursor cell expansion. "We had previously demonstrated that a 2D culture system was more efficient than traditionally used 3D culture systems for endothelial precursor cell expansion1," notes Jain. "However, it was not known if 2D culture system is also superior for iPS cell derived cells. We therefore systematically compared 2D and 3D culture methods for the generation of endothelial cells from hiPS cells, since a growing body of evidence suggests that embryonic stem cells and iPS cells do not necessarily behave in the same way. Interestingly," he continues, "we found that while 3D culture systems generated well-characterized endothelial cells in vitro, the 3D-endothelial cells derived from iPS cells did poorly in vivo. Indeed, we found that the 2D culture system – similar to our previous experience with derivation of endothelial cells from human embryonic stem cells – was capable of generating long-lasting functional blood vessels in mice in conjunction with the CD34, NP1 and KDR triple marker selection."
Jain points out that several methods of generating endothelial cells or perivascular cells in vitro have been previously reported, and that a variety of techniques could be used to differentiate iPS cells into endothelial cells that are comparable in vitro – but most of them do not last when implanted in animals. "Until our study," Jain says, "the outstanding issue remained the demonstration of iPS derived blood vessel functionality due potentially to a lack of vasculogenic cells functionally competent in vivo in addition to the methodological challenges to study engineered vessel function. To our knowledge, this study is the first to successfully demonstrate long-lasting blood vessels derived from hiPS cells."
Fukumura also points out that both endothelial cells and mesenchymal precursors are critical to establishing stable vasculature – and that the Edwin L. Steele Laboratory showed this for the first time in vivo when they previously created functional vasculature using an engineered construct of human umbilical endothelial cells (HUVECs) co-implanted with mouse embryonic stem cells2. "These vessels lasted for a year, or normal animal life span, in the cranial window models of mice" Fukumura recounts. "What is unique in the current study is the use of a side population during the selection of endothelial cells precursors as a source of MPCs. In this way, we can use the same hiPS cells for both endothelial cell and MPC generation – a technique that is extremely important for future clinical application in that it will reduce in vitro culture burden by half."
In order to allow the possibility of translational potential in the future, Fukumura continues, both endothelial and mesenchymal progenitor cell types needed for successful formation of durable blood vessels must be of human origin. "Adult somatic tissue derived vascular precursors have limited proliferative capacity," he explains, "and the use of vascular precursors from human embryonic stem cells pose ethical and immunological concerns that are averted with the use of iPS-derived vasculogenic cells." Since most animal studies of vasculogenic cells are conducted for periods of a few weeks to a couple of months, they thereby fall short of showing the durability and normal functionality required for the application of these procedures. "Our goal was to extend this period to several months in order to demonstrate not just vasculogenic potential of iPS-derived vascular precursors, but also the durability and functionality of the engineered vessels."
Regarding these myriad challenges, Samuel tells Medical Xpress that the discovery of a method to induce pluripotency in mature cells – that is to bring back a cell from a differentiated state to a stem-like state – has brought enormous potential to the field of cell-based regenerative medicine. "However," she adds, "the challenge remains to derive functional progeny from these so-called induced pluripotent stem (iPS) cells. In this study we demonstrated efficient generation of vascular precursor cells from iPS cells in vitro that were vasculogenic in vivo, and were able to create engineered vessel constructs from iPS cells derived both from healthy and diseased – that is, diabetic – donors."
Samuel further states that once safety issues of iPS-based cellular therapy have been resolved, these results provide proof-of-principle that autologous (where the same individual organism is both donor and recipient) hiPS-cell-derived vascular precursors could be used for in vivo applications. "We used retroviral and lentiviral techniques to deliver a combination of transcription factors that are known to generate iPS cells. Moreover, we developed endothelial and mesenchymal progenitor cell vascular precursors from both healthy controls and Type 1 Diabetic patients."
However, Jain explains, in order to be used in a clinic environment, the scientists will need to use non-integrating methods of reprogramming; the iPS-derived vascular precursors would have to be evaluated in detail to understand the human host immune response to the introduction of iPS-derived vascular precursors; and long term follow-up would be required out to ensure that there are no adverse effects. "We've shown that, once optimized, iPS derived blood vessels are durable in vivo in mice. This suggests that our approach of generating iPS-derived blood vessels has a potential to be translated in the clinic in conditions requiring vascular development such as cardiovascular disease, or revascularization in long-standing ischemic, arterial, diabetic or non-healing ulcers.'
Having established a robust technique of generating blood vessels from iPS cells derived from healthy donors, the researchers next wanted to evaluate blood vessel forming ability of vasculogenic cells generated from iPS cells derived from Type 1 Diabetic (T1D) patients in whom vascular complications occur. "Our study validates that endothelial and mesenchymal precursor cells derived from T1D-iPS cells were able to form functional blood vessels in vivo," Fukumura adds. "Additionally, the T1D-iPS cell derived blood vessels were not apparently different morphologically and functionally from those of healthy iPS-derived blood vessels."
Looking ahead, Fukumura describes other innovations they might develop and apply to the current experimental design:
- Establish safe iPS cell generation protocols using non-viral, non-integrating methods that would ensure zero-foot print technology
- Understand the human host immune response to introduction of iPS derived vascular precursors and long term follow up to ensure that there are no adverse effects
- Understand the factors underlying the variability in iPS cell line differentiation into vascular progeny in healthy controls and patient specific iPS cell lines
- Develop tailor made endothelium specific to target organs
- Establish large animal models of disease to examine effects of iPS-blood vessels that may more closely recapitulate humans than mice
In addition, Samuel outlines next steps the scientists are planning for their research. "We'll examine the mechanisms that influence what confers vasculogenic potential of iPS-derived vascular precursors," she says. "Our plan is to use other patient specific cell lines from vascular disease to derive vascular progeny that would lead to understanding pathophysiology of disease."
Jain also sees other areas of research that might benefit from their study. "The ability to regenerate or repair blood vessels could make a crucial difference in the treatment of cardiovascular disease and other conditions caused by blood vessel damage, such as the vascular diabetic complications particularly found in Type 2 Diabetes. Furthermore," he concludes, "these iPS-derived vasculogenic cells have broad application for regenerative medicine as blood vessels are essential for any functional organs as recently shown in the generation of functional liver in mice with co-implantation of hepatocytes, mesenchymal stem cells and vascular endothelial cells."
More information: Generation of functionally competent and durable engineered blood vessels from human induced pluripotent stem cells, PNAS published online before print July 16, 2013, doi:10.1073/pnas.1310675110
Related
1Endothelial cells derived from human embryonic stem cells form durable blood vessels in vivo, Nature Biotechnology 25, 317-318 (2007), doi:10.1038/nbt1287 (PDF)
2Tissue engineering: Creation of long-lasting blood vessels, Nature 428, 138-139 (11 March 2004), doi:10.1038/428138a (PDF)
© 2013 Medical Xpress. All rights reserved.