First to measure the concerted activity of a neuronal circuit
Neurobiologists from the Friedrich Miescher Institute for Biomedical Research have been the first to measure the concerted activity of a neuronal circuit in the retina as it extracts information about a moving object. With their novel and powerful approach they can now not only visualize networks of neurons but can also measure functional aspects. These insights are direly needed for a better understanding of the processes in the brain in health and disease.
For many decades electrophysiology and genetics have been the main tools in the toolbox of approaches to study individual neurons in the central nervous system to understand perception and behavior. In the last five years however, neurobiology has been riding a wave of technological advances that brought unprecedented insights: Optogenetics and genetically encoded activity sensors has allowed scientists to control and measure the activity of clearly defined neurons; the application of rabies viruses enabled the visualization of networks of interconnected nerve cells. What was still missing, was the link between neural circuit and monitoring of activity.
Scientists from the Friedrich Miescher Institute for Biomedical Research have now been the first to measure the concerted activity of a neuronal circuit in the retina as it extracts information about the movement of an object.
In a world defined through eyesight, it is crucial to be able to discern whether something moves towards us, moves away or moves next to us. It comes as no surprise then that in the retina several parallel neuronal circuits are reserved for the extraction of information about movement and that most of them are dedicated to the analysis of the direction of motion.
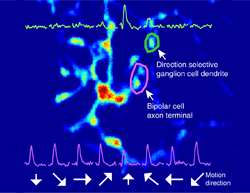
As they report online in Neuron, Keisuke Yonehara and Karl Farrow, two Postdoctoral Fellows in Botond Roska's team at the FMI, have now been able to monitor the activity of all circuit elements in a motion sensitive retinal circuit at once, and pinpoint the site, at a subcellular level, where the information about the direction of the movement becomes encoded. To achieve this, they used genetically altered rabies viruses expressing calcium sensors developed by the laboratory of Klaus Conzelmann in Munich. The special property of rabies viruses is that they move across connected neurons and therefore are able to deliver the sensors to all circuit elements within a defined neuronal circuit. Simultaneous two-photon imaging allowed them then to monitor activity in every part of the neuronal circuit at once, even in subcellular compartments, such as axons, synapses and dendrites.
"We are extremely thrilled that with this new method, which combines the power of genetically altered rabies viruses with very powerful two-photon microscopy, we are now able to link circuit architecture with activity and ultimately function," comments Yonehara. "We have illustrated the power of the method for a better understanding of the perception of movement and are convinced that the method will allow us to reach a better understanding of many processes in the retina and in other parts of the brain."
More information: Yonehara, K. et al. (2013) The first stage of cardinal direction selectivity is localized to the dendrites of retinal ganglion cells, Neuron.
Tang, J. et al. (2013) A nanobody-based system using fluorescent proteins as scaffolds for cell-specific gene manipulation. Cell 154:928-939. www.ncbi.nlm.nih.gov/pubmed/23953120