September 12, 2013 report
The thermodynamics of thought: Soliton spikes and Heimburg-Jackson pulses
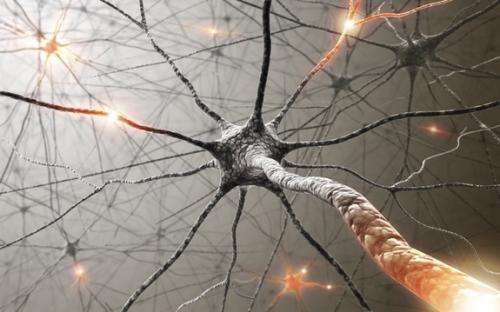
(Medical Xpress)—In the familiar rendering of a neuron, as in the image above, the so-called electrical spikes are usually depicted as short pulses. In reality, if the spike lasts for over a millisecond and its expanding front travels at 100 meter/second, then we are in fact talking about a physical disturbance that would extend some 10cm. Insofar as artists are informed by real neuroscience, and not fantasy, it also becomes necessary to insure that neuroscience is informed by real physics. In a recent discussion of brain activity maps, we made ample mention of the fact that this electrical disturbance is a multiphysical event whose full experimental signature is only partially accounted for by current models. A mounting body evidence now suggests an alternative, and more fundamental explanation—neurons and their membranes in particular, send signals to each other using sound.
The commonly accepted view of spikes is that they are propagated by resistive ion currents dissipatively flowing through channel pores. One glaring inconsistency with this model is that actual measurements have repeatedly shown that while heat is liberated during the initial phase of the pulse, it is immediately reabsorbed in its entirety in the second phase. In other words, the heat production integrated over the duration of the pulse is zero. This implies that an adiabatic and reversible process, rather then a dissipative one is operating in nerves. Many of the early heat and pressure measurements on nerves were actually done by the founding fathers, more or less, of the electrical theory. If you read their early explorations into the electrical activity of nerves, you will find that they repeatedly warned against over interpreting their own empirical data fits and models to the exclusion of more general thermodynamic models.
An intriguing new theory, originally put forth by Heimburg and Jackson at the Neils Bohr Institute in Copenhagen, holds that neurons communicate using finely-tuned mechanical pulses known as solitons. The important feature of these membrane-based waves is that they propagate with very little change in shape or loss of energy. This should come as welcome news for those who have struggled to understand how neurons could possibly have enough energy to do all that they seem to do. The prevailing electrical theory prescribes a voracious ATP appetite to the ion pumps which are needed to make things happen. While it is possible for the ATP neuroaccountants to balance the pump budget by juggling various aerobic and anaerobic sources, doing so leaves little ATP for anything else.
Heimburg and Jackson have done extensive analysis, both experimental and theoretical, of how membranes undergo phase transitions to create solitons. They have found that many variables, including hydrostatic pressure, pH, calcium concentration and membrane proteins all have well defined effects on membrane melting temperature, which in turn determines its excitability. Among the many testable predictions of their theory is its potential to explain some curious experimental phenomena related to anesthesia. In particular, they observe that anesthetics invariably lower the melting point of membranes while hydrostatic pressure increases it due to the latent volume changes. Experimentally, these concepts are supported by the fact that deeply anesthetized tadpoles can be quickly returned to normal activity when subjected to a pressure of 50 bars. Tadpoles are often used in this kind of research because there is an easy-to-understand criteria: they are considered anesthetized when half of them become inactive and sink to the ground.
In an effort to help bridge some of these ideas to skeptical neurobiologists, I challenged Dr. Heimburg with some real world, potentially soliton-collapsing questions. Theoretically, there could be many fly-in-the-ointment scenarios that could obstruct soliton propagation. For example, when an invertebrate, or a cold blooded animal like a frog, jumps into cold water, the thermodynamics of its membranes may need to be adjusted for survival in this environment. The full variety and speed of lipid or other adaptations is not yet known, but may be approached experimentally. Heimburg notes that spikes can be elicited by cooling, and his model also predicts that slight heating should have an inhibitory effect on spikes. In other settings, we should note here that researchers have been able to activate neurons by an IR laser illumination, presumably through a heating mechanism. Clearly, the full picture of the thermodynamics of neuron activation is not yet in hand, but it seem likely that this seemingly contradictory effect must be occurring within different temperature regimes altogether.
Other potential threats to solitons propagating on long axons in different environments might be imagined. While Heimburg has extensively modeled soliton collisions, their behavior at the many branch points found as an axon undergoes 10-fold fractal bifurcation is as yet unexplored. Just as in an electrical system, impedance matching and potential reflection at branch points needs to be considered for the mechanical component as well. Perhaps the greatest mismatch would occur at the thousand or so synaptic terminations where the remaining energy of soliton can potentially be apportioned in various ways between transmission, dissipation, and reflection, depending on the mechanical impedance of the synaptic cleft.
The problems with the current theory of signal transmission in neurons does not end at the axon, but extends all the way to the synapse. It has been known for some time that diffusion alone cannot govern the discharge of transmitter if the measured timecourse of events is to hold sway. Various stopgaps including pressurized vesicles exercising propulsive release of their contents into protein constrained extracellular channels have been imagined to help speed things up, but here, the energy-efficient soliton model has significant potential to illuminate. The key specializations at the synapse that have evolved for vesicle fusion and transmitter release clearly contribute to its growth and transformation. However, as alluded to above, the raw energetic requirements to even try to match spikes with fusions at the observed release probabilities of less than one-half at each synapse, continue to boggle not only the neuroaccountants, but also those who attempt to define any familiar semblance of computation for this seemingly fickle architecture.
Phase transitions in lipids have previously been shown to be important in other areas of cell function. For example, the formation of membrane "rafts" are now known to play a role in protein sorting in the endoplasmic reticulum and golgi bodies. Researchers including Matthias Schneider at Boston University, have gotten involved to conduct their own measurements on the thermodynamic properties of lipids. In particular, they have recently studied mechanical pulse propagation and relaxation in worm vessels and nerves. They have also begun to look at opto-mechanical coupling at lipid interfaces using fluorescent dyes, and similarly, acoustical effects on proteins and vesicles.
Solitons have been found in a wide variety of different systems. Within neuroscientific applications, they have already come to be known as Heimburg-Jackson pulses. The literature is still fairly complex and it tends to revolve around precise measurements in neurons to determine things like enthalpy and compressibility. Expanding the scope of soliton propagation in membranes to include the real biologic particulars of things like membrane-attached cytoskeletal elements and myelination will be critical to gaining more widespread acceptance for the theory. Bringing more biologists into the field may help us to better understand exactly how and when cells may have hit upon this fascinating capability—and put it to use.
More information: 1. www.plosone.org/article/fetchO … ject.action?uri=info%3Adoi%2F10.1371%2Fjournal.pone.0066773&representation=PDF
© 2013 Medical Xpress