Single molecule shown to regulate emotional behavior of mice

Mood and emotion are extremely complex aspects of behavior that are known to involve the neurotransmitters serotonin and norepinephrine. Both neurotransmitters are broken down by an enzyme called monoamine oxidase A (MAO-A), and drugs that interfere with this system, such as the anti-depressant fluoxetine (Prozac), have long been used to treat mood disorders. Jun Aruga and colleagues from the RIKEN Brain Science Institute in Wako have now shown that a ligase enzyme called Rines regulates MAO-A activity and could prove to be a promising therapeutic target for the treatment of such disorders.
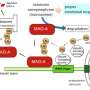
The metabolism of MAO-A, like that of many other proteins in the cell, is regulated by what is known as the ubiquitin–proteasome pathway, which tags old, misfolded or otherwise unwanted molecules and dispatches them to a cellular trash can. Aruga and his colleagues hypothesized that this pathway would be critical for regulating MAO-A levels in the brain.
To investigate the role of this pathway, the researchers used genetic engineering to create mice lacking the gene encoding the Rines E3 ubiquitin ligase—an enzyme that determines which proteins will be tagged for destruction.
Deletion of the Rines gene had a dramatic effect on the emotional behavior of the mice. For example, mice lacking the gene were far more reluctant to explore a new environment and spent less time in open spaces compared to healthy mice, indicating that the mutant mice were more anxious and had an abnormal stress response.
The researchers then examined the brains of these mice and found that the altered emotional behaviors were associated with significantly reduced levels of serotonin and norepinephrine in the locus ceruleus, prefrontal cortex and amygdala—regions of the brain that regulate emotion and stress responses. This was accompanied by enhanced activity of MAO-A in the locus ceruleus, the main source of norepinephrine in the brain. The researchers also found that some of the abnormal emotional behaviors were abolished by MAO inhibitors.
"The next step is to clarify the change in emotional response abnormalities using the animals' personal history," says Aruga. "Studies in humans indicate that the prevalence of aggressive and antisocial behavior in adults with the low-level MAO-A variant is affected by their history of stress during childhood, so personal history and gene–environment interaction studies with the mutant mice would contribute to a more comprehensive understanding of the pathophysiology of aggression and antisocial behavior."
More information: Kabayama, M., Sakoori, K., Yamada, K., Ornthanalai, V. G., Ota, M., Morimura, N., Katayama, K., Murphy, N. P. & Aruga, J. Rines E3 ubiquitin ligase regulates MAO-A levels and emotional responses, The Journal of Neuroscience 33, 12940–12953 (2013). dx.doi.org/10.1523/JNEUROSCI.5717-12.2013