Animal study reveals sex-specific patterns of recovery from newborn brain injury
Physicians have long known that oxygen deprivation to the brain around the time of birth causes worse damage in boys than girls. Now a study by researchers from the Johns Hopkins Children's Center conducted in mice reveals one possible reason behind this gender disparity and points to gender-specific mechanisms of brain repair following such injury.
The results of the study, to appear in the February issue of the journal Neuroscience, show that inherent differences in the way newborn brains react to the sex hormone estradiol may be behind the sex-specific response to brain damage and cell repair.
"Our observations reveal intriguing differences in the way male and female brains respond to injury following oxygen deprivation and in the manner in which they recover following such injury," says lead investigator Raul Chavez-Valdez, M.D., a neonatologist at the Johns Hopkins Children's Center.
In addition, the researchers say, neurons in male and female brains undergo different type of cell death following oxygen deprivation that may be due to the presence of certain receptors that trigger sex-specific pathways of cell demise.
Lastly, the scientists say, their results clarify an earlier observation that the brains of male mice, while sustaining worse damage overall, tend to respond better to certain types of therapies that halt neuronal cell death.
The findings, Chavez-Valdez says, underscore the need to explore questions about gender differences in all studies, including those conducted in animals, infants and children. Answering these questions in this case could prove to be a stepping stone toward finding precisely targeted, gender-based therapies to stimulate brain cell preservation and recovery, the team says.
"Our findings show just how important gender-specific research is. Not only are sex differences a powerful player in the pathology and course of disease, but our results indicate that such differences begin to emerge very early in life, in the very first days of birth and, indeed, perhaps long before that," says senior study investigator Frances Northington, M.D., a neonatologist at the Johns Hopkins Children's Center.
For their experiments, the investigators homed in on a critical cell repair protein called brain-derived neurotrophic factor (BDNF), known for its role in stimulating the growth and regeneration of neurons in the brain. Adequate amounts of this neuron-nurturing protein ensure cell health in areas of the brain associated with a range of vital functions, such as processing of sensory information, learning and memory.
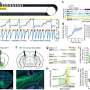
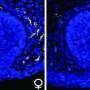
Examining tissue from newborn mice with brain injury, the researchers noticed that following oxygen-deprivation, cells rapidly release BDNF, causing a spike in its levels, followed by a precipitous dip 96 hours thereafter. The team observed that BDNF levels in male and female mice followed the same spike-dip patterns. However, they found a disproportionately higher presence of two BDNF receptors in the brains of female mice that promote a milder form of cell death after oxygen deprivation. These receptors, the researchers say, trigger a form of neuronal death known as apoptosis, a type of programmed cell death. The brains of male mice, on the other hand, had fewer of these injury-blunting receptors. The scarcity of such receptors in male mice, the researchers believe, causes neurons in the male brain to undergo necrosis, a more violent type of cell death marked by bursting or disintegration of the cell, which can also wreak damage on neighboring cells.
When researchers treated brain-injured animals with a substance called necrostatin-1, or nec-1, previously shown to halt necrotic cell death in the brains of mice, they noted a markedly different response to treatment in male and female animals. The brains of male mice had 41 percent more BDNF than female mice 96 hours after injury. In other words, nec-1 exhibited sex-specific protective effects. Could sex hormones explain this gender gap?
To answer this question, the researchers turned their attention to estradiol, the chief female sex hormone, also found in smaller amounts in males. Newborn male and female mice had similar levels of estradiol in their brains, the researchers noted, yet, they somehow responded differently to it. The investigators observed that following treatment with nec-1, the brain cells of male mice had a higher concentration of a receptor known as alpha estrogen receptor. Alpha estrogen receptors' primary role is to increase cell sensitivity to estradiol, a type of estrogen, but one of its lesser known actions is to promote BDNF production. Thus, the researchers say, nec-1 appears to fuel the expression of such receptors in the male brain, which in turn trigger more BDNF production.
Investigators say the Neurosciences Intensive Care Nursery team at Johns Hopkins is also planning a study in human newborns to track the behavior of BDNF in response to brain injury and treatments.
Temporary cutoff of oxygen to the brain before, during or immediately after birth can cause a range of neurologic, developmental and learning disorders, including cerebral palsy, which is believed to occur in one to three out of 1,000 full-term newborns. Newborn boys have a 40 percent greater risk of developing cerebral palsy following hypoxic brain injury.