October 7, 2014 report
A closer look at the blood-brain barrier
(Medical Xpress)—Thousands of people today have various kinds of stimulators placed deep in their brains in the hope of curing their ills. Many others require systems of tubes, catheters, and shunts penetrating deep into their brain ventricles to deliver chemotherapy, analgesics, or to drain overpressurized cerebral liquids. Invariably, as in the commonly used Ommaya reservior for example, these devices have a crude and bulky insertion that is simply plowed through healthy brain tissue, sparing little in its path. To develop more delicate instrumentation for the brain we must imagine working with its natural barriers and protections rather than against them. In other words, we must come to know the wisping membranes that balance the brain's fluid constitution, and the blood brain barrier that seals its vessels, as we would know our own skin.
Viruses, bacteria, fungi, parasitic tapeworms, or even cancer cells all have their own unique tricks for bypassing the blood-brain barrier (BBB). Whether it is brute force penetration, deception, chemical stealth, or sheer overwhelming numbers, they will find a way in if the infection persists long enough. As the march towards industrially-scaled electronic miniaturization penetrates into various neuromarkets, it is inevitable that researchers will begin to see the BBB-busting skillsets of microbes applied to artificial devices for the brain. While we don't typically think of inserting recording or stimulating electrodes in the brain via the vasculature, it is perhaps the least invasive method to enable the fantastic hypothetical technologies—Smart Dust or fiber optic-based optogenetics for example—that await us.
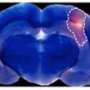
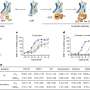
To actually see this mysterious BBB, MRI images can be taken after the injection of paramagnetic gadolinium contrast agents into the bloodstream. These compounds generally stay within the cerebral vasculature and only leak out where the permeability of the BBB has altered from its natural state of health. Low concentrations of Gadolinium are typically prefered in these kinds of tests because of potential toxicity. A study just published in the journal Nanoscale suggests there may be something better. By using superparamagnetic iron oxide particles encapsulated with inert hydrophilic polyethylene glycol (PEG), these researchers were able to obtain MRI images of the BBB as it dynamically responds to assault.
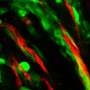
The researchers call their carefully-engineered nanoparticles PEGylated SPIONs (Superparamagnetic Iron Oxide Nanoparticles). These SPIONs proved particularly adept at enhancing T2-weighted images of the permeabilized BBB. Compared to the T1-weighted gadolinium scans, the T2-weighted scans can give greater detail in imaging various brain pathologies. The researchers also were able to analyze the SPION label within the brain parenchyma itself. "Parenchyma" is a catch-all term meaning the visceral or function parts of an organ. For the brain, that generally means the neurons and glial cells. We know, however, that there is much more to the brain than this. By some estimates, with over 100,000 miles of astrocytic endfeet-lined capillary tubing in its fractal coastline, the BBB and other membranous ventricular linings might be considered an organ itself.
A brief trip through the history of the BBB may be an illuminating exercise for us here. One-hundred years ago this year, the BBB was discovered when scientists were trying to find ways to get drugs into the brain to treat African trypanosomiasis (sleeping sickness). Paul Ehrlich developed the first successful treatments against the African trypanosomes that caused the disease. He coined the terms "chemotherapy" and "magic bullet" after the trypan dyes which could kill the parasite's cells while sparing the healthy cells.
Seeing and killing are not completely isolate events in this context. When injected into the bloodstream, it was known that trypan dyes would not label the brain, thereby highlighting some kind of border. However, it was not known if this was because of a barrier or because it simply did not stick to the brain. Injecting the dye directly into the nether hollows of the brain (which labelled it) revealed that it must be the former. Trypan dyes also happen to make good indicators of cell viability because they generally do not pass through intact membranes. There are a few exceptions though.
One thing that directly emerged from the study of trypanosomes is a core principle in cell biology and neuroscience known as endocytosis. Gaining passage through membrane boundaries by budding in through in vesicles is a key event in infection. The control of secretory trafficking through vesicles passaging into and out of the brain is as important as that which occurs ubiquitously within the brain. Some foreign assailants, like rabies, hijack axonal transport machinery to their own nefarious ends. Incredibly, the rabies virus (which travels retrogradely in the same circle as endocytosed growth factor receptors) somehow manages to speed the whole process up so that it actually travels faster than the endogenous flow.
The endothelial cells which line the BBB are similar to those found in any other organ yet are held to a slightly higher standard. The junctions they make are tighter, and subject to more points of control. There is also a sparse layer of pericytes above the endothelial cells, and above them are the tiling hands of astrocytes. Doctors today have a few crude tricks they can use to manipulate the BBB. They can administer mannose, for example, to osmotically pull fluid from the brain during hydrocephalus. There are now signs that things may soon become more a little technically advanced than mannose and Ommaya shunts.
Non-invasive measurements of flow and pressure will be essential to understanding what is happening inside the brain when it has been altered by natural or by artificial means. One company, Ornim Medical, is now offering instruments to record parameters through the skull within the micro-capillary bed. By measuring near IR light that has been modulated by ultrasound within the tissue through the acousto-optic effect they can directly gauge perfusion. These and other techniques may prove critical once we begin to heavily implant our brains with assistive technologies—and more critically perhaps, have medical issues with them.
More information: Magnetic Resonance Imaging of Post-Ischemic Blood-Brain Barrier Damaging with PEGylated Iron Oxide Nanoparticles, Nanoscale, 2014, Accepted Manuscript, DOI: 10.1039/C4NR03942D
Abstract
Blood-brain barrier (BBB) damaging during ischemia may induce devastating consequences like cerebral edema and hemorrhagic transformation. This study presents a novel strategy for dynamically imaging of BBB damaging with PEGylated supermagnetic iron oxide nanoparticles (SPIONs) as contrast agents. The employment of SPIONs as contrast agents made it possible to dynamically image the BBB permeability alterations and ischemic lesions simultaneously with T2-weighted MRI, and the monitor could last up to 24 h with a single administration of PEGylated SPIONs in vivo. The ability of the PEGylated SPIONs to highlight BBB damaging by MRI was demonstrated by the colocalization of PEGylated SPIONs with Gd-DTPA after intravenous injection of SPION-PEG/Gd-DTPA into a mouse. The immunohischemical staining also confirmed the leakage of SPION-PEG from cerebral vessels into parenchyma. This study provides a novel and convenient route for imaging BBB alteration in the experimental ischemic stroke model.
© 2014 Medical Xpress